Abstract
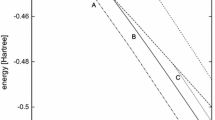
It was shown by Claverie that the interactions between atoms and molecules make unphysical electronic solutions of the Schradinger equation accessible in perturbation calculations of intermolecular interactions, accessible in the sense that the perturbation expansion is likely to converge to an unphysical solution if it converges at all. This is a difficult problem because there are generally an infinite number of unphysical states with energies below that of the physical ground state. We have carried out configuration interaction calculations on LiH of both physical and unphysical states. They show that avoided crossings occur between physical and unphysical energy levels as the interaction between the two atoms is turned on, i.e. as the expansion parameter λ is increased from 0 to 1. The avoided crossing for the lowest energy state occurs for λ < 0.8, implying that the perturbation expansion will diverge for larger values of λ. The behavior of the energy levels as functions of λ. is shown to be understandable in terms of a two-state model. In the remainder of the paper, we concentrate on designing effective Hamiltonians which have physical solutions identical to those of the Schrödinger equation, but which have no unphysical states of lower energy than the physical ground state. We find that we must incorporate ideas from the Hirschfelder-Silbey perturbation theory, as modified by Polymeropoulos and Adams, to arrive finally at an effective Hamiltonian which promises to have the desired properties, namely, that all unphysical states be higher in energy than the physical bound states, that the first and higher order corrections to the energy vanish in the limitR = ∞. that the leading terms of the asymptotic 1/R expansion of the energy be given correctly in second order, and that the overlap between the zeroth order wave function and the corresponding eigenfunction of the effective Hamiltonian be close to one.
Similar content being viewed by others
References
P. Claverie, Theory of intermolecule forces. I. On the inadequacy of the usual Rayleigh-Schrödinger perturbation method for the treatment of intermolecular forces, Int. J. Quant. Chem. 5 (1971)273.
J.D. Morgan and B. Simon, Behavior of molecular potential energy curves for large nuclear separations, Int. J. Quant. Chem. 17 (1980)1143.
W.H. Adams, Perturbation theory of intermolecular interactions: What is the problem, are there solutions?, Int. J. Quant. Chem. S24 (1990)531.
J.O. Hirschfelder and R. Silbey, New type of molecular perturbation treatment, J. Chem. Phys. 45 (1966)2188.
J.O. Hirschfelder, Perturbation theory for exchange forces II, Chem. Phys. Lett. 1 (1967)363.
E.E. Polymeropoulos and W.H. Adams, Exchange perturbation theory. III. Hirschfelder-Silbey type, Phys. Rev. A17 (1978)24.
R. Peierls, Perturbation theory for projected states, Proc. Roy. Soc. London A333 (1973)157;
B. Atalay, A. Mann and R. Peierls, Perturbation theory for projected states. II. Convergence criteria and a soluble model, Proc. Roy. Soc. London A335 (1973)251.
E.E. Polymeropoulos and W.H. Adams, Exchange perturbation theory. II. Eisenschitz-London type, Phys. Rev. A17 (1978)18.
R. Ahlrichs, Convergence properties of the intermolecular force series (I/R-expansion), Chem. Phys. Lett. 41 (1976)7.
W. Kutzelnigg, The primitive wave function in the theory of intermolecular interactions, J. Chem. Phys. 73 (1980)343.
P.R. Certain, J.O. Hirschfelder, W. Kolos and L. Wolniewicz, Exchange and Coulomb energy of H2 determined by various perturbation methods, J. Chem. Phys. 49 (1968)24.
D.M. Chipman and J.O. Hirschfelder, Perturbation theories for the calculation of molecular interaction energies. I. General formalism, J. Chem. Phys. 59 (1973)2830.
P.O. Löwdin, On the non-orthogonality problem connected with the use of atomic wave functions in the theory of molecules and crystals, J. Chem. Phys. 18 (1950)365.
A.C. Wahl, P.J. Bertocini and R.H. Land, BISON: A FORTRAN computer system for the calculation of analytic self-consistent-field wave functions, properties, and charge densities for diatomic molecules; Part I. User's manual and general program description, Argonne National Laboratory Report 7271, Argonne, IL (1968).
W.H. Adams, M.M. Clayton and E.E. Polymeropoulos, Perturbation calculations of the interaction energy between two hydrogen atoms in their ground states, Int. J. Quant. Chem, S18 (1984)393.
A. Mann and V. Privman, Localized symmetry-adapted perturbation theory and a new tight-binding expansion, Phys. Rev. Lett. 49 (1982)1068; Pseudosymmetry — a new method of deriving perturbation expansions, Chem. Phys. Lett. 106(1984)447.
W.H. Adams, On the solution of the Schubdinger equation in terms of wave functions least distorted from products of atomic wave functions, Chem. Phys. Lett. 11 (1971)441.
W.H. Adams, On the solution of the Schubdinger equation for Hz in terms of a wave function least distorted from a product of atomic wave functions, Int. J. Quant. Chem. S7 (1973)127.
W.H. Adams and C.A. Venanzi, unpublished calculations (1980).
W.H. Adams, Perturbation theory of intermolecule interactions: Are second order Rayleigh-Schrödinger energies meaningful?, Int. J. Quant. Chem. S25 (1991)165.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Adams, W.H. The problem of unphysical states in the theory of intermolecular interactions. J Math Chem 10, 1–23 (1992). https://doi.org/10.1007/BF01169168
Issue Date:
DOI: https://doi.org/10.1007/BF01169168