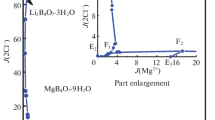
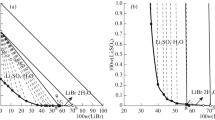
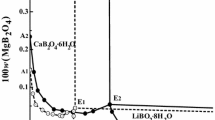
Summary
-
1.
Ternary aqueous systems containing lithium chloride and chlorides of Group II metals have been studied by the isothermal method.
-
2.
In the systems LiCl-BeCl2-H2O, LiCl-CaCl2-H2O, LiCl-SrCl2-H2O and LiCl-BaCl2-H2O, no chemical compounds are formed between the components.
-
3.
In the systems LiCl-ZnCl2-H2O and LiCl-CdCl2-H2O, the chemical compounds ZnCl2·2LiCl·2,5H2O and LiCl·CdCl2·2,5H2O have been found.
-
4.
In the system LiCl-HgCl2-H2O the solubility of HgCl2 is enhanced in presence of LiCl. No chemical compounds between the components were found.
Similar content being viewed by others
Literature cited
N. K. Voskresenskaya and O. K. Yanatyeva Bull. Acad. Sci. USSR (Chemical Series), 1937, No. 1, 97.
H. Basset and J. Sandersen, J. Chem. Soc., 1932, 1855.
H. Benraht, Z. anorg. Chem., 21, 145 (1934).
A. V. Novoselova and I. G. Sosnovskaya, J. Gen. Chem., 21, 813 (1951).
J. Banks, Analyst, 77, 918 (1952).
I. M. Korenman, Quantitative Microchemical Analysis, State Chem. Press, Moscow, 1949, p. 233.
V. P. Blidin, Proc. Acad. Sci. USSR, 88, No. 3, 457 (1953).
Schmidt, Ann. Chim., (10), 1, 367 (1929.
W. Herz and Paul, Z. anorg. Chem. 82, 4 (1913).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bildin, V.P. Heterogenous equilibria in systems containing lithium chloride and chlorides of group II metals. Russ Chem Bull 3, 337–345 (1954). https://doi.org/10.1007/BF01167808
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01167808