Summary
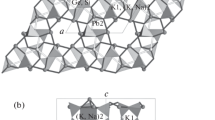
The crystal structure of the tetragonal fumarole mineral nabokoite, Cu7TeO4(SO4)5 · KCl (a=9.833 (1).Å,c=20.591(2) Å, space groupP 4/ncc, Z=4) was determined by single crystal X-ray methods on type material from Kamchatka. The structure contains complicated {[CU7TeO4(SO4)4]SO4} sheets that are intercalated by K and Cl ions. Nabokoite presents the first example of a Te(IV)O4 pyramid with exactly tetragonal symmetry. The K ions have a somewhat unusual, rather flat coordination.
Zusammenfassung
Die Kristallstruktur des tetragonalen Fumarolenminerals Nabokoit, Cu7TeO4(SO4)5 · KCl (a=9,833(1)Å,c=20,591(2)Å, RaumgruppeP 4/ncc, Z=4) wurde an Typ-Material von Kamtschatka mit Röntgen-Einkristallmethoden bestimmt. Die Struktur enthält komplizierte [Cu7TeO4(SO4)4]SO4-Schichten, die durch K- und Cl-Ionen verbunden werden. Nabokoit liefert das erste Beispiel für eine Te(IV)O4-Pyramide mit exakt tetragonaler Symmetrie. Die K-Ionen haben eine etwas ungewöhnliche, ziemlich flache Koordination.
Similar content being viewed by others
References
Bayer G (1969) Zur Kristallchemie des Tellurs. Telluroxide und Oxidverbindungen mit Tellur. Fortschr Mineral 46: 42–72
Cocco G, Fanfani L, Zanazzi PF (1972) Potassium. Crystal chemistry. “Handbook of Geochemistry” Vol II/2 (Ed:Wedepohl KH). Springer, Berlin Heidelberg New York: 19-A-1-19-A-9
Effenberger H (1985) Synthesis and crystal structure of Na5Cu3(PO4)2(PO4HPO4). Z Kristallogr 172: 97–104
—— (1987) The structure of pentasodium tricopper(II) hydrogentetraarsenate(V). Acta Cryst C 43: 399–402
rdInternational Tables for X-Ray Crystallography” Vol IV (1974) (Ed:Ibers JA, Hamilton WC). The Kynoch Press: Birmingham
Koçak M, Platte C, Trömel M (1979) Bariumhexaoxoditellurat (IV, VI): Sauerstoffkoordinationszahl Fünf am vierwertigen Tellur. Acta Cryst B 35: 1439–1441
Naboko CI, Glavatskikh CF (1980) Exhalative copper ore formation in the posteruptive phase of the Great Fissure Eruption of Tolbachik. Vukanologija u Seismologija No 4: 50–64 (in Russian)
——, —— (1983) Posteruptive metasomatism and ore formation. Nauka: Moscow (in Russian)
Popova VI, Popov VA, Rudashevsky NS, Glavatskikh SF, Polyakov VO, Bushmakin AF (1987) Nabokoite, Cu7TeO4(SO4)5 · KCl, and atlasovite, Cu6Fe3+Bi3+O4(SO4)5 · KCl—new minerals of volcanic exhalations. Zap Vses Mineral Obshch 116: 358–367 (in Russian)
Trömel M (1980) Empirische Beziehungen zur Sauerstoffkoordination um Antimon(III) und Tellur(IV) in Antimoniten und Telluriten. J Solid State Chem 35: 90–98
Vergasova LP, Filatov SK (1983) The new mineral tolbachite. Doklady Akad Nauk SSSR 270: 415–417 (in Russian)
——, ——Serafimova EK, Starova GL (1984) Piypite—a new mineral of volcanic sublimates. Doklady Akad Nauk SSSR 275: 714–717 (in Russian)
Watkins SF, Fronczek FR, Wheelock KS, Goodrich RG, Hamilton WO, Johnson WW (1988) Structure of Y2BaCuO5. Acta Cryst C44: 3–6
Wells AF (1949) The crystal structure of atacamite and the crystal chemistry of cupric compounds. Acta Cryst 2: 175–180
—— (1984) Structural Inorganic Chemistry. Clarendon Press, Oxford
Wuensch BJ (1972) Sulfur. Crystal chemistry. “Handbook of Geochemistry” Vol II/2 (Ed:Wedepohl KH). Springer, Berlin Heidelberg New York: 16-A-1-16-A-19
Zemann J (1961) Die Kristallchemie des Kupfer. Fortschr Mineral 39: 59–68
—— (1968) The crystal chemistry of the tellurium oxide and tellurium oxosalt minerals. Z Kristallogr 127: 319–326
—— (1971) Zur Stereochemie des Te(IV) gegenüber Sauerstoff. Monatsh Chemie 102: 1209–1216
—— (1972) Copper. Crystal Chemistry. In:Wedepohl KH (ed) Handbook of geochemistry, Vol II/3. Springer, Berlin Heidelberg New York, 29-A-1-29-A-13
—— (1974) Tellurium. Crystal Chemistry. In:Wedepohl KH (ed) Handbook of geochemistry, Vol 11/4. Springer, Berlin Heidelberg New York, 52-A-1-52-A-5
Author information
Authors and Affiliations
Additional information
With 2 Figures
Rights and permissions
About this article
Cite this article
Pertlik, F., Zemann, J. The crystal structure of nabokoite, Cu7TeO4(SO4)5 · KCl: The first example of a Te(IV)O4 pyramid with exactly tetragonal symmetry. Mineralogy and Petrology 38, 291–298 (1988). https://doi.org/10.1007/BF01167095
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01167095