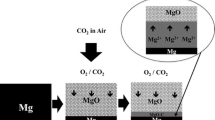
Conclusions
-
1.
At 750° no protective film preventing evaporation of magnesium is formed on the surface of a molten magnesium alloy of the Mg−Al−Zn system in an atmosphere of nitrogen or N2+1–20 vol. % SO2.
-
2.
In an atmosphere of 40–50 vol. % N2+60–50 vol. % CO2 a coating of soot is formed on the surface of the molten magnesium alloy, preventing evaporation of magnesium.
-
3.
Sulfur dioxide slows down the oxidation of magnesium in an atmosphere of CO2, and 20 vol. % SO2 is sufficient to block defects in the MgO film.
-
4.
In an atmosphere of CO2 with 1–10 vol. % SF6 a dense protective film is formed on the surface of magnesium, the thickness of the film changing little with the SF6 concentration.
-
5.
The optimal quantity of SF6 in a nitrogen atmosphere is 1.5–3 vol. %; the thickest and densest protective film is formed on the surface of the molten magnesium alloy under these conditions.
Similar content being viewed by others
Literature cited
J. Hanawalt, Gieserrei-Prax., No. 9, 143 (1975).
M. Boussion, R. Darras, and D. Leclercq, Rev. Metall.,56, 61 (1959).
I. Leontis and F. Rhines, Trans. AIME,166, 285 (1946).
S. Gregg and W. Jepson, J. Inst. Met.,87, 187 (1959).
A. Schneider and U. Esch. Z. Metallk.,32, 173.
Additional information
Moscow Aviation Technology Institute. Translated from Metallovedenie i Termicheskaya Obrabotka Metallov, No. 3, pp. 52–55, March, 1977.
Rights and permissions
About this article
Cite this article
Aleksandrova, Y.P., Roshchina, I.N. Interaction of magnesium with gases. Met Sci Heat Treat 19, 218–221 (1977). https://doi.org/10.1007/BF01167005
Issue Date:
DOI: https://doi.org/10.1007/BF01167005