Abstract
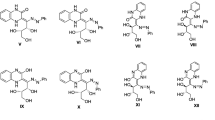
PMR spectroscopy was used to show that the hydrazones of (2-benzothiazolyl) and (2-benzoxazolyl)acetic acids exist in solution as equilibrium mixtures of stereoisomeric forms due to conformational and geometric isomerism.
Similar content being viewed by others
References
I. P. Bezhan, V. A. Khrustalev, K. N. Zelenin, and B. P. Nikolaev, Zh. Org. Khim.,14, 754 (1978).
K. N. Zelenin, V. V. Pinson, A. A. Potekhin, I. P. Bezhan, V. A. Khrustalev,and P. S. Lobanov, Zh. Org. Khim.,14, 490 (1978).
S. I. Yakimovich, V. N. Nikolaev, and O. A. Afonina, Zh. Org. Khim.,15, 922 (1979).
A. I. Rutavichyus and S. P. S. P. Valyulene, Trudy Akad. Nauk Lithuanian SSR, Ser. B, 4(155), 61 (1986).
V. G. Yusupov, S. I. Yakimovich, S. D. Nasirdinov, and S. D. Parpiev, Zh. Org. Khim.,16, 415 (1980).
Additional information
Institute of Chemistry, 2600 Vilnius, Lithuania. Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 5, pp. 708-712, May, 1995. Original article submitted April 3, 1995.
Rights and permissions
About this article
Cite this article
Rutavlcius, A., Valiulene, S. & Kuodis, Z. Isomerism of hydrazones of (2-benzothiazolylthio)- and (2-benzoxazolylthio)acetic acids. Chem Heterocycl Compd 31, 629–633 (1995). https://doi.org/10.1007/BF01166343
Issue Date:
DOI: https://doi.org/10.1007/BF01166343