Abstract
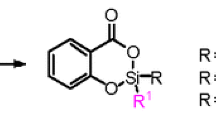
Tetracoordinated derivatives of 2-halo-2-aryl-5H-1.2-oxatelluroles and 1-halo-l-butyl-3H-benz-2.1-oxatelluroles were synthesized by the oxidation of 3-aryltelluro-2 propen-l-ols and the dehydrogenation of 2-butyldihalotellurobenzyl alcohols. The halogen atoms in these compounds are readily replaced in nucleophilic substitution reactions. The reaction of benzoxatelluroles with bromine is accompanied by cleavage of O-Te bonds and leads, depending on the nature of the substituent at C(3), to 2-(butylbromotelluro)benzaldehyde or 2-(butyldibromotelluro)benzophenone.
Similar content being viewed by others
References
I. D. Sadekov, G. M. Abakarov, E. I. Sadekova, and V. I. Minkin, Sulfur Reports,6, 15 (1986).
I. D. Sadekov and B. B. Rivkin, Khim. Geterotsikl. Soedin., No. 3, 291 (1991).
M. R. Derry, B. J. Murray, and D. L. Smith, J. Am. Chem. Soc.,105, 875 (1983).
M. R. Detty and B. J. Murray, J. Am. Chem. Soc.,105, 883 (1983).
I. D. Sadekov, A. V. Zakharov, and B. B. Rivkin, Zh. Org. Khim., in print.
A. A. Maksimenko, I. D. Sadekov, and V. I. Minkin, Zh. Obshch. Khim.,60, 471 (1990).
S. Uemura, S. Fukuzawa, and S. Patil, J. Organometall. Chem.,243, 9 (1983).
K. Ohe, H. Takahashi, S. Uemura, and N. Sugita, J. Org. Chem.,52, 4859 (1987).
S. R. Buzilova, L. I. Vereshchagin, I. D. Sadekov, and V. I. Minkin, Zh. Obshch. Khim.,46, 932 (1976).
S. R. Buzilova, I. D. Sadekov, T. V. Lipovich, T. M. Fillipova, and L. I. Vereshchagin, Zh. Obshch. Khim.,47, 1999 (1977).
A. Gordon and R. Ford, Chemist's Handbook [Russian translation], Mir, Moscow (1976), p.302.
V. I. Minkin, I. D. Sadekov, A. A. Maksimenko, O. E. Kotran, and Yu. T. Struchkov, J. Organometall. Chem.,402, 331 (1991).
W. R. McWhinnie, Phosphorus, Sulfur and Silicon and Relat. Elem.,67, 107 (1992).
S. Uemura and S. Fukuzawa, Tetrahedron Lett.,23, 1181 (1982).
S. M. Barros, M. J. Dabdoub, and J. V. Comasseto, Organometallics,8, 1661 (1989).
I. D. Sadekov, A. A. Maksimenko, and V. I. Minkin, Khim. Geterotsikl. Soedin., No. 1, 122 (1981).
R. S. Michalak, S. R. Wilson, and J. C. Martin, J. Am. Chem. Soc.,106, 7529 (1984).
L. J. Adzima and J. C. Martin, J. Org. Chem.,42, 4006 (1977).
V. I. Minkin and R. M. Minyaev, Zh. Org. Khim.,11, 1993 (1975).
K. Chikamatsu, T. Otsubo, F. Ogura, and H. Yamaguchi, Chem. Lett., No. 11, 1081 (1982).
I. D. Sadekov, A. A. Maksimenko, and V. I. Minkin, Chemistry of Organotellurium Compounds [in Russian], Izd. Rostovsk. Gos. Univ., Rostov-on-the-Don (1983).
J. L. Piette and M. Renson, Bull. Soc. Chim. Belg.,80, 669 (1971).
J. M. Talbot, M. Renson, and J. L. Piette, Bull. Soc. Chim. France, No. 1, 294 (1976).
Z. Arnold and J. Zemblicka, Coll. Czech. Chem. Commun.,24, 2385 (1959).
G. T. Morgan and R. E. Kellett, J. Chem. Soc., No. 1, 1080 (1926).
G. T. Morgan and H. D. K. Drew, J. Chem. Soc., No. 2, 2307 (1925).
J. L. Piette and M. Renson, Bull. Soc. Chim. Belg.,79, 367 (1970).
I. D. Sadekov, A. A. Maksimenko, and B. B. Rivkin, Zh. Org. Khim.,19, 616 (1983).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 2, pp. 266–279, February, 1994. Original article submitted December 22, 1993.
Rights and permissions
About this article
Cite this article
Sadekov, I.D., Maksimenko, A.A., Zakharov, A.V. et al. 5H-1,2-oxatelluroles and their benzo analogs: Synthesis and reactions. Chem Heterocycl Compd 30, 243–253 (1994). https://doi.org/10.1007/BF01165022
Issue Date:
DOI: https://doi.org/10.1007/BF01165022