Abstract
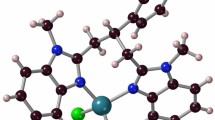
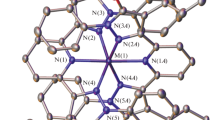
The results of an X-ray structure determination of one of the three possible chloride salts of [Cr(en) F2(OH2)2]+ show the two fluoro ligands to betrans to each other. The purple crystals are monoclinic in the space groupC2/c-C 62h , witha=11.409(3),b=9.319(2),c=10.260(3) Å,β=128.73(2)° andD c =1.73 g cm−3 forZ=4. Least-squares refinement based on 1341 independent observed reflections resulted in a finalR of 0.026. The molecule lies on a crystallographic two-fold axis, and the Cr-F, Cr-N, and Cr-O bond lengths are 1.885(1), 2.044(2), and 1.997(2) Å respectively. It is suggested that the ill-defined shoulder in the visible electronic spectrum of this complex is indicative of atrans dihalo geometry.
Similar content being viewed by others
References
Bang, E., and Pederson, E. (1978)Acta. Chem. Scand. Ser. A, 833.
Brencic, J. V., and Leban, I. (1981)Z. Anorg. Allg. Chem. 480, 213.
Cromer, D. T., and Mann, J. L. (1968)Acta Crystallogr. A 24, 341.
Hamilton, W. C., and Ibers, J. C.,Hydrogen Bonding in Solids, (W. A. Benjamin, New York, 1968), p. 16.
House, D. A., and Garner, C. S. (1966)Inorg. Chem. 5, 840.
House, D. A. Hughes, R. G., and Garner, C. S. (1967)Inorg. Chem. 6, 1077.
Hughes, R. G., and Garner, C. S. (1968)Inorg. Chem. 7, 1988.
International Tables for X-Ray Crystallography (1974) Vol. IV (Kynoch Press, Birmingham, England), pp. 149–150.
Pyke, S., and Linck, R. G. (1971)Inorg. Chem. 10, 2445.
Stromberg, R., and Larking, I. (1969)Acta. Chem. Scand. 23, 343.
Vaughn, J. W. (1980) unpublished data.
Vaughn, J. W. (1981)Inorg. Chem. 20, 2397.
Vaughn, J. W., and Marzowski, J. (1973)Inorg. Chem. 12, 2346.
Vaughn, J. W., and Seiler, G. J. (1979)Synth. React. Inorg. Met. Org. Chem. 9, 1.
Vaughn, J. W., and Yeoman, A. M. (1976)Inorg. Chem. 15, 2320.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vaughn, J.W. Fluoro-containing complexes of chromium (III). II. Crystal structure oftrans-difluorodiaquo-ethylenediaminechromium(III) chloride. Journal of Crystallographic and Spectroscopic Research 13, 231–239 (1983). https://doi.org/10.1007/BF01161517
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01161517