Abstract
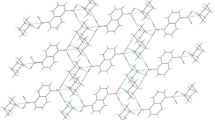
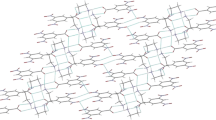
1,8-bis(dimethylamino)naphthalene hydrobromide dihydrate, C14H19N +2 Br · 2H2O, crystallizes in the space groupPnma witha=7.264(1),b=11.794(1),c=18.986 (2) Å,Z=4,D m =1.34 g cm−3,D X =1350 kg cm−3. The structure was determined by Patterson and Fourier methods and refined to anR factor of 0.038. The 1,8-bis(dimethylamino)naphthalene cation has mirror symmetry, with the mirror plane passing along the C(9)-C(10) bond. The two N atoms are moved slightly out of the naphthalene plane. Theperi interactions are discussed in relation to aromaticity and to the N-C (ring) bond lengths. The structure contains channels of H bonds (Br− and water molecules) parallel toa which alternate with channels of cations in theb andc directions. One of the water molecules is disordered.
Similar content being viewed by others
References
Alder, R. W., Bowman, P. S., Steele, W. R. S., and Winterman, D. R. (1968)Chem. Commun., 723.
Anderson, J. E., Franck, R. W., and Mandella, W. L. (1972)J. Am. Chem. Soc. 94, 4608.
Balasubramaniyan, V. (1966)Chem. Rev. 66, 567.
Cruickshank, D. W. J. (1957)Acta Cryst. 10, 504.
Cruickshank, D. W. J. (1962)Tetrahedron 17, 155.
Einspahr, H., Robert, J. B., Marsh, R. E., and Roberts, J. D. (1973)Acta Cryst. B 29, 1611.
Jaskólski, M. (1979) Ph.D. Thesis, A. Mickiewicz University, Poznań, Poland.
Kruszewski, J., and Krygowski, T. M. (1972)Tetrahedron Lett., 3839.
Krygowski, T. M., and Kruszewski, J. (1973)Bull. Acad. Pol. Sci. Ser. Sci. Chim. 21, 409.
Olovsson, I., and Jonsson, P.-G. (1976)The Hydrogen Bond, Vol. 2 (North-Holland, Amsterdam, New York, Oxford), p. 433.
Truter, M. R., and Vickery, B. L. (1972)J. Chem. Soc. Dalton Trans., 395.
Wong-Ng, W., Nyburg, S. C., Awwal, A., Jankie, R., and Kresge, A. J. (1982)Acta Cryst. B 38, 559.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pyżalska, D., Pyżalski, R. & Borowiak, T. Structure of 1,8-bis(dimethylamino)naphthalene hydrobromide dihydrate. Journal of Crystallographic and Spectroscopic Research 13, 211–220 (1983). https://doi.org/10.1007/BF01161515
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01161515