Abstract
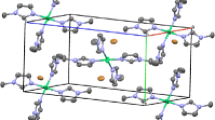
[NBu4][Br3] interacts with C6H6 to form an air-stable liquid clathrate. The tribromide anion reacts with phenol to give para-bromophenol in>95% yield. The HBr which is generated in the reaction combines with the Br− anion to form [Br-H-Br]−. The latter forms the basis for a new liquid clathrate, [NBu4][Br-H-Br]·n C6H6. The parent salt, [NBu4][Br3], crystallizes in the monoclinic space group C2/c witha=12.983(5),b=10.380(7),c=16.222(6) Å,β=93.93(3)°, andD c =1.47 g cm−3 forZ=4. The final R value is 0.068 based on 787 observed reflections.
Similar content being viewed by others
References
Atwood, J. L. (1982)U. S. Patent 4,321,127.
Atwood, J. L. (1984)Inclusion Compounds. J. L. Atwood, J. E. D. Davies, and D. D. MacNicol, (eds.) (Academic Press, London), Vol. 1, pp. 375–405.
Atwood, J. L. (1986)Chemical Separations, C. J. King and J. D. Navratil (eds.) (Litarvan, Arvada, Colorado), pp. 335–356.
Berthelot, J., Guette, C., Ochefoune, M., Desbene, P.-L., and Basselier, J.-J. (1986)J. Chem. Res. (S), 381.
Bogaard, M. P., and Rae, A. D. (1982)Cryst. Struct. Commun. 11, 175.
Bruce, M. I., Humphrey, M. G., Koutsantonis, G. A., and Nicholson, B. K. (1982)J. Organomet. Chem. 296, C47.
Buckles, R. E., Popov, A. I., Zelezny, W. F., and Smith, R. J. (1951)J. Am. Chem. Soc. 73, 4525.
Cotton, F. A., Lewis, G. E., and Schwoizer, W. (1986)Inorg. Chem. 25, 3528.
Emsley, J. (1980)Chem. Soc. Rev. 9, 91.
Germain, G., Main, P., and Woolfson, M. M. (1971)Acta Crystallogr. A27, 368.
Holton, J., Lappert, M. F., Ballard, D. G. H., Pearce, R., Atwood, J. L., and Hunter, W. E. (1979)J. Chem. Soc., Dalton Trans. 45.
Means, C. M. (1988) Dissertation, University of Alabama.
Rogers, R. D., Baker, J. C., and Atwood, J. L. (1984)J. Cryst. Spec. Res. 14, 333.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Coleman, A.W., Mitchell Means, C., Bott, S.G. et al. Air-stable liquid clathrates. 1. Crystal structure of [NBu4][Br3] and reactivity of the [NBu4][Br3]·5 C6H6 liquid clathrate. Journal of Crystallographic and Spectroscopic Research 20, 199–201 (1990). https://doi.org/10.1007/BF01160976
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01160976