Abstract
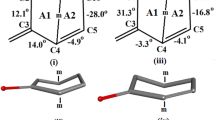
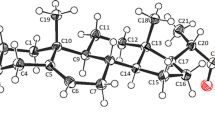
The structure of the title compound, C21H30O3, was determined by X-rays.M r =330.5, orthorhombic,P212121,a=8.4451(9),b=10.7074(8),c=20.1525(40) Å,V=1822(8) Å3,Z=4,D x =1.205 mg m−3. CuKα radiation (1.54184 Å),μ(CuKα)=5.85 cm−1,F(000)=720. FinalR=0.054R w =0.049 for 1774 unique reflections. The structure was solved usingMultan. TheA ring adopts an intermediate sofa-half-chair conformation and is bent toward theα face of the steroid skeleton. RingsB andC have typical chair conformations, and theD ring has a slightly distorted half-chair conformation. The progesterone side chain has a typical conformation, and the C16-C17-C20-O20 torsion angle is −17.9°. An intermolecular hydrogen bond is formed between the hydroxyl group and the progesterone side chain.
Similar content being viewed by others
References
Allen, F. H., Kennard, O., and Taylor, R. (1983) The systematic analysis of structural data as a research tool in organic chemistry,Acc. Chem. Res. 16, 146.
Campsteyn, H., Dupont, L., and Dideberg, O. (1972)Acta Crystallogr. B 28, 3032–3041.
Campsteyn, H., Dupont, L., Dideberg, O., and Mandel, N. (1973)Acta Crystallogr. B 29, 1727.
Duax, W. L., and Norton, D. A. (1975)Atlas of Steroid Structure, Vol. 1 (Plenum Press, New York).
Duax, W. L., Griffin, J. F., Rohrer, D. C., Weeks, C. M., and Ebright, R. H. (1984)Biochemical Actions of Hormones, Vol. XI (Academic Press, New York), pp. 187–205.
Griffin, J. F., Duax, M. L., and Weeks, G. M. (1984)Atlas of Steroid Structure, Vol. 2 (Plenum Press, New York).
Johnson, C. K. (1970)Ortep, A Fortran Thermal-ellipsoid Program for Crystal Structure Illustrations, ORNL-3794 (Oak Ridge National Laboratory, Oak Ridge, Tennessee).
Main, P., Hull, S. E., Lessinger, L., Germain, G., Declercq, J.-P., and Woolsfon, M. M. (1980)Multan 80, A system of computer programs for automatic solution of crystal structures from X-ray diffraction data (Universities of York/England/Louvain, Belgium).
Motherwell, W. D. S. (1976)Pluto. A program for plotting molecular and crystal structures (University Chemical Laboratory, Cambridge, England).
Rohrer, D. C., and Smith, G. D. (1980)Fitmol in Prophet Molecules. W. Rindone and A. Kush (eds.) (Bolt Beranek and Newman, Cambridge, Mass.).
Sheldrick, C. M. (1976)Shelx76. A program for crystal structure determination (University Chemical Laboratory, Cambridge, England). Spilman C. H., Wilks, J. W., Beuving, D. C., and Forbes, K. K. (1979) Technical Report (The Upjohn Company), p. 7.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gałdecki, Z., Grochulski, P. & Wawrzak, Z. Structure of 11α-hydroxy-progesterone, C21H30O3 . Journal of Crystallographic and Spectroscopic Research 20, 145–148 (1990). https://doi.org/10.1007/BF01160966
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01160966