Abstract
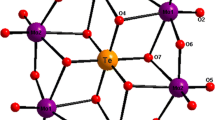
Oxidation of Mo2[S2CN(C2H5)2]6 with iodine in a 1∶2 molar ratio yielded the complex MO[S2CN(C2H5)2]4 I3, for which magnetic susceptibility, NMR, IR, UV/visible spectra, and single crystal X-ray structure are reported. Crystals of Mo[S2CN(C2H5)2]4 I3 are monoclinic, space groupC2/c (No. 15),a=26.242(7),b=7.003(1),c=20.447(3) Å,D o =1.97,Z=4,D c =1.890 g cm−3. The structure was determined from 1909 observed reflections and refined by blocked cascade least squares to anR factor of 0.042. The core of the cation, Mo[S2CN(C2H5)2] +4 , is dodecahedral with approximate point symmetryD 2d (¯42m). The eight S atoms coordinated to the Mo center can be divided into two sets, each forming a distorted tetrahedron. The set constituting a flattened tetrahedron has a mean Mo-S distance of 2.545(2) Å, whereas the set that forms an elongated one has a mean value of 2.500(2) Å. The oxidation state of +5 for the molybdenum atom is supported by the thioureide band [v(RNC)] at 1507 cm−1, which lies between those of similar Mo(IV) and Mo(VI) complexes.
Similar content being viewed by others
References
Bonati, F., and Ugo, R. (1967)J. Organomet. Chem. 10, 257.
Brinkhoff, H. C., Cras, J. A., Steggerda, J. T., and Willemes, J. (1969)Recl. Trav. Chim. Pays-Bas 88, 633.
Brown, D. A., Gordon, B. J., Glass, W. K., and O'Daly, C. J. (1972)Proc. Xlth Intern, Conf. Coord. Chem., Toronto, p. 646.
Brown, D. A., Glass, W. K., and Jasim, K. S. (1982)Proc. Climax Fourth Intern. Conf. on the Chemistry and Uses of Molybdenum Climax Molybdenum Co., Ann Arbor, Michigan, p. 156.
De Titta, G. T., Edmonds., J. W., Langs, D. A., and Hauptman, H. (1975)Acta Crystallogr,31, 472.
Diamond, R. (1969)Acta Crystallogr., Sect. A 25, 43.
Garner, C. D., Howlader, N. C., Mabbs, F. E., MaPhail, A. T., Miller, R. W., and Onan, K. D. (1978)J. Chem. Soc., Dalton Trans., 1582.
International Tables for X-ray Crystallography (1974) Vol. 4 (Kynoch Press, Birmingham, England), pp. 99, 149.
Jasim, K. S. (1982) Ph.D. Thesis, National University of Ireland.
Jorgensen, C. K. (1962)J. Inorg. Nucl. Chem. 24, 1571.
Kopfmann, G., and Huber, R. (1968)Acta Crystallogr., Sect. A 24, 348.
Mak, T. C. W., Jasim, K. S., and Chieh, C. (1984)Angew. Chem. Int. Ed. Engl. 23, 391.
McGarvey, B. R. (1966)Inorg. Chem. 5, 476.
McAuliffe, C. A., and Sayle, B. J. (1978)Inorg. Chim. Acta 30, 35.
Mitchell, P. C. H., and Scarle, R. D. (1975)J. Chem. Soc. Dalton Trans., 110.
Moore, F. W., and Larson, M. L. (1967)Inorg. Chem. 6, 998.
Nieuwpoort, A. (1974)J. Less Common. Met. 36, 271.
Nieuwpoort, A., and Staggerda, J. T. (1976)Recl. Trav. Chim. Pays-Bas 95, 250.
North, A. C. T., Philips, D. C., and Mathews, F. S. (1968)Acta Crystallogr., Sect. A 24, 351.
Schilling, J. W. (1970) InCrystallographic Computing, F. R. Ahmed, ed. (Munksgaard, Copenhagen), p. 201.
Sheldrick, G. M. (1982), InComputational Crystallography, D. Sayre, ed. (Oxford University Press, New York), p. 506.
van der Aalsvoort, J. G. M., and Beurskens, P. T. (1974)Cryst. Struct. Commun. 3, 653.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jasim, K.S., Umbach, G.B., Chieh, C. et al. Structural and spectroscopic study of tetrakis(N,N-diethyldithiocarbamato)molybdenum(V) triiodide, Mo[S2CN(C2H5)2]4 I3 . Journal of Crystallographic and Spectroscopic Research 15, 271–280 (1985). https://doi.org/10.1007/BF01160487
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01160487