Abstract
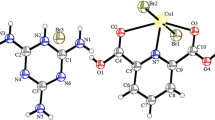
From an equimolar mixture of guanidine and maleic acid, two different crystal modifications of guanidinium hydrogen maleate, CH6N +3 ·C4H3O −4 , were isolated and their crystal structures determined using three-dimensional diffractometer data. Crystals of both forms are orthorhombic:Pca21,a=19.012(8)b=3.758(1),c=11.119(5) Å,Z=4 for form I;Fdd2,a=18.505(2),b=20.007(3),c=8.406(2) Å,Z=16 for form II. The structures were solved by direct methods and refined by full-matrix least squares to conventionalR values of 0.064 for form I and 0.043 for form II, with 488 and 605 reflections respectively. The structures consist of planar guanidinium cations C(NH2) +3 and hydrogen-maleate residues C4H3O −4 . There are no significant differences in distances and angles within the guanidinium and hydrogen-maleate units. However, a substantial difference appears in the length and geometry of the intramolecular hydrogen bond O⋯H⋯O. The packing of cations and anions is also completely different in the two forms. Infrared and Raman spectra of the title compound are presented, and salient differences between them and the spectrum of the symmetrically bonded potassium hydrogen maleate are discussed briefly.
Similar content being viewed by others
References
Adams, J. M. (1978)Acta Cryst. B 34, 1218.
Adams, J. M., and Ramdas, V. (1976)Acta Cryst. B 32, 3224.
Adams, J. M., and Ramdas, V. (1978)Acta Cryst. B 34, 2150.
Avbelj, F., Hadži, D., and Orel, B. (1984a).Spectrochim. Acta A, in press.
Avbelj, F., Hodošček, M., and Hadži, D. (1984b).Spectrochim. Acta A, in press.
Cramer, D. T., and Mann, J. B. (1968)Acta Cryst. B 28, 3632.
Darlow, S. F., and Cochran, W. (1961)Acta Cryst. 14, 1250.
Ellison, R. D., and Levy, H. A. (1965)Acta Cryst. 19, 260.
Golič, L., and Leban, I. (1982)Croat. Chem. Acta 55, 41.
Gupta, M. P., Prasad, S. M., and Yadav, B. (1972)Cryst. Struct. Commun. 1, 211.
Gupta, M. P., Prasad, S. M., and Gupta, T. N. T. (1975)Acta Cryst. B 31, 37.
Hadži, D., Orel, B., and Novak, A. (1973)Spectrochim. Acta A 29, 1745.
Hamilton, W. C. (1959)Acta Cryst. 12, 609.
Hsu, Bennett, and Schlemper, E. O. (1978)Acta Cryst. B 34, 930.
Hsu, Bennett, and Schlemper, E. O. (1980)Acta Cryst. B 36, 3017.
James, M. N. G., and Matsushima, M. (1976)Acta Cryst. B 32, 1708.
Maillols, J., Bardet, L., and Marignan, R. (1969a)J. Chim. Phys. 66, 522.
Maillols, J., Bardet, L., and Marignan, R. (1969b)J. Chim. Phys. 66, 529.
Main, P., Woolfson, M. M., Lessinger, L., Germain, G., and Declercq, J. P. (1974)Multan 74. A system of Computer Programs for Automatic Solution of Crystal Structures. Universities of York, England and Louvain-la-Neuve, Belgium.
Murthy, A. S. N., Bhat, S. N., and Rao, C. N. R. (1970)J. Chem. Soc. A, 1251.
Orel, B., and Hadži, D. (1977)Chim. Scr. 11, 102.
Prout, C. K., Carruthers, J. R., and Rossotti, F. J. C. (1971)J. Chem. Soc. A, 3342.
Sheldrick, G. M. (1976)Shelx-76. A program for Crystal Structure Determination. University of Cambridge, England.
Stewart, R. F., Davidson, E. R., and Simpson, W. T. (1965)J. Chem. Phys. 42, 3175.
Tomkinson, J., Braid, I. J., Howard, J., and Waddington, T. C. (1982)Chem. Phys. 64, 151.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Golič, L., Leban, I., Detoni, S. et al. X-ray diffraction and vibrational spectra of the guanidine:maleic acid (1:1) complex—two crystal forms. Journal of Crystallographic and Spectroscopic Research 15, 215–228 (1985). https://doi.org/10.1007/BF01160482
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01160482