Abstract
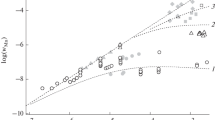
The authors examine some of the regularities displayed when UO2 is oxidized by manganese dioxide in sulfuric acid solution, both without and with added iron ions. It is shown that the redoX potential indicates only the potential capacity of the system, and is not an unambiguous criterion of the degree of oxidation of UO2; the effective reaction rate is limited by the reaction mechanism.
A study is made of the way in which the degree of oxidation and dissolution of UO2, at 20 and 85°C in sulfuric acid solution, depends On the redox potential when MnO2 and Fe (II) are added. The relation is expressed by an S-shaped curve. It is shown that, during oxidation of UO2. by manganese dioxide in sulfuric acid solution, iron ions act as a catalyst.
Similar content being viewed by others
Literature Cited
O. F. Klochko, V. P. Zobnin, Sb. trudov GIPKha, vyp. 4, 171 (1958).
M. S. Zakhar'evskii, Some Problems of Redox Potential Measurement: Report given to the scientific conference on pH measurement [in Russian] (Tbilisi, 1960).
V. I. Ksezenko and A. S. Stasinevich, Technology of Bromine and Iodine [in Russian] (Goskhimizdat, Moscow, 1960).
W. Eskenfelder, Instrum. and Automat.,27, 1082 (1954).
S. Dayton, Mining World,19, 36 (1957).
A. Gaudin et al., J. Metals,8, 1065 (1956).
J. Clegg and D. Foley, Uranium Ore Processing (Massachusetts, USA, 1958).
R. Bock and M. Hermann, Z. anorg. und allgem. Chem.,273, 1 (1953).
W. M. Latimer, Oxidation State of the Elements and their Potentials in Aqueous Solutions [Russian translation] (IL, Moscow, 1954).
V. G. Khlopin and A. M. Gurevich, Izv. AN SSSR, Otd. khim. nauk, No. 4, 271 (1943).
D. M. Chizhikov and G. S. Balikhina, Tsvetnye metally, No. 9, 67 (1936).
E. A. Kanevskii and V. A. Pchelkin, Atomnaya énergiya,10, 138 (1961).
E. A. Kanevskii and A. P. Filippov, Radiokhimiya,5, 602, 741 (1963).
T. Arden, Industr. Chemist,32, 376, 202 (1956).
J. Arthur and R. Wheeler, J. South African Inst. of Mining and Metallurgy,57, 631 (1957).
A. Thunaes, Canad. Mining J.,77, 123 (1956).
Vikt. I. Spitsyn et al., Atomnaya énergiya,8, 261 (1960).
Additional information
Translated from Atomnaya Énergiya, Vol. 17, No. 3, pp. 205–208, September, 1964
Rights and permissions
About this article
Cite this article
Filippov, A.P., Kanevskii, E.A. Reduction-oxidation potential and degree of extraction of uranium in sulfuric acid solutions. At Energy 17, 925–928 (1964). https://doi.org/10.1007/BF01125282
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01125282