Abstract
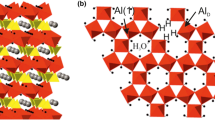
The effect of lithium nitrate mineralizer on the thermal stability and phase transformations of kaolinite has been studied using7Li,27Al and29Si magic angle spinning (MAS) nuclear magnetic resonance (NMR) in tandem with Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD) and TEM. As the temperature is raised, lithium nitrate melts, wets the surface of the kaolinite particles, and Li+ diffuses into the bulk of the crystals. The mineralizer retards the dehydroxylation of kaolinite by 15–20 °C.27Al MAS NMR indicates that metakaolinite originating from mineralized kaolinite contains less five-coordinated and slightly more four-coordinated Al. This promotes the transformation of metakaolinite into hightemperature phases which contain only four- and six-coordinated Al. Thus at 920 °C, mineralized samples (MS) containγ-alumina type spinel but non-mineralized samples (NMS) do not. At higher temperatures, MS and NMS contain different amounts of spinel phase, mullite and cristobalite.29Si MAS NMR shows that cristobalite crystallizes from segregated amorphous silica. The process is reversed between 1300 and 1400 °C in MS (but not in NMS) and cristobalite vitrifies. This is probably caused by the lowering of the liquid temperature and subsequent rapid quenching of the melt.
Similar content being viewed by others
References
C. W. Parmelee andA. R. Rodriguez,J. Am. Ceram. Soc. 25 (1942) 1.
S. P. Chaudhuri,Trans. Brit. Ceram. Soc. 28 (1969) 24.
J. Lemaitre andB. Delmon,J. Mater. Sci. 12 (1977) 2056.
D. N. Hinckley, in “Clays and Clay Minerals, Proceedings of the 13th National Conference”, Madison, Wisconsin, 1964, edited by W. F. Bradley and S. W. Bailey (Pergamon Press, New York, 1963) pp. 220–35.
K. Srikrishna, G. Thomas, R. Martinez, M. P. Corral, S. Aza andJ. S. Moya,J. Mater. Sci. 25 (1990) 607.
J. Rocha andJ. Klinowski,Phys. Chem. Min. 17 (1990) 179.
H. J. Percival, J. F. Duncan andP. K. Foster,J. Amer. Ceram. Soc. 57 (1974) 57.
J. Rocha andJ. Klinowski,Angew. Chim. 29 (1990) 553.
J. F. Lambert, W. S. Millman andJ. J. Fripiat,J. Amer. Chem. Soc. 111 (1989) 3517.
J. Sanz, A. Madani, J. M. Serratosa andS. Aza,J. Amer. Ceram. Soc. 71 (1988) C-418.
G. Engelhardt andD. Michel, “High-resolution solid-state NMR of silicates and zeolites” (Wiley, New York, 1987).
E. Göbel, W. Müller-Warmuth andH. Olyschläger,J. Magn. Reson. 36 (1979) 371.
M. Bulens andB. Delmon,Bull. Soc. Chim. Belg. 86 (1977) 405.
M. Bulens, A. Leonard andB. Delmon,J. Amer. Ceram. Soc. 61 (1978) 81.
C. S. F. Gomes,Miner. Petrogr. Acta 29A (1985) 381.
A. G. Verduch andJ. S. M. Corral, in “Proceedings of the International Clay Conference”, Madrid (1972) p. 131.
L. Heller-Kallai,Clay Min. 13 (1978) 221.
S. A. T. Redfern,ibid. 22 (1987) 447.
A. R. West, “Solid-state chemistry and its applications” (Wiley, Chichester, 1984).
R. B. Sosman, “The phases of silica” (Rutgers University Press, New Brunswick, 1965).
G. H. Frischat, “Ionic diffusion in oxide glasses: Diffusion and defect monographs series”, 3/4 (Trans Tech, Aedermannsdorf, Switzerland, 1975).
I. W. M. Brown, K. J. D. Mackenzie, M. E. Bowden andR. H. Meinhold,J. Am. Ceram. Soc. 68 (1985) 298.
ACS, “Phase diagrams for ceramists”, compiled at the National Bureau of Standards (The American Ceramic So- ciety, Ohio, 1969).
A. Paul, “Chemistry of glasses” (Chapman and Hall, London, New York, 1982).
Y. Kubo andY. Yamabe, “Proceedings of the International Clay Conference”, Tokyo, Vol. 1 (1969) p. 915.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rocha, J., Klinowski, J. & Adams, J.M. Solid-state NMR elucidation of the role of mineralizers in the thermal stability and phase transformations of kaolinite. J Mater Sci 26, 3009–3018 (1991). https://doi.org/10.1007/BF01124836
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01124836