Abstract
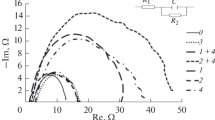
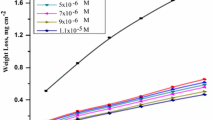
The inhibition effect of potassium iodide on the corrosion of stainless steel has been examined using weight loss and electrochemical measurements. Potassium iodide was found to be an excellent inhibitor for stainless steel types 316, 430 and 440 (efficiency more than 95%), and moderate for type 304. The corrosion rates in 1m HCl and in presence of potassium iodide fit an Arrhenius equation. The presence of potassium iodide lowered both the activation energy and the rate of reaction. Polarization measurements indicated that potassium iodide acts as an anodic inhibitor particularly at higher concentrations and as a cathodic inhibitor at lower concentrations. The synergistic effect of potassium iodide on the inhibitive efficiency of the organic substances was studied.
Similar content being viewed by others
References
G. Schmitt,Br. Corros. J. 19 (1984) 165.
N. D. Tamashov, “Theory of corrosion and protection of metals” (Macmillan, New York, 1966).
Dhirendra, G. N. Pandey andB. Sanyal,Corrosion Prevention Control 27(3) (1980) 13.
Viorel Branzoi, Ahmed Kabir andMihai Ciopec,Rev. Roum. Chim. 31 (1986) 571.
Shashi Gupta, Dhirendra, B. Sanyal andG. N. Pandey,Corrosion Prevention Control 28 (1981) 21.
Gentaro Kano andTutuk Subowo,Mem. Fac. Engng Fukui Univ. 34(1) (1986) 8.
Gentaro Kano, Kiyoshi Horita andHartati Soeroso,ibid. 35(2) (1987) 213.
Szkluka-Smialowsta,Z. Corr. Trait. Prot. Fin. 17 (1969) 283.
W. McLeod andR. R. Rogers,Mater. Prot. 5(12) (1966) 28.
N. Hackerman, E. S. Snavely andJ. S. Payne,J. Electrochem. Soc. 113 (1966) 677.
M. G. Fontana andR. W. Stachle, “Advances in Corrosion Science and Technology”, Vol. I Plenum Press, (London, New York, 1970).
O. L. Riggs Jr,Corrosion 24 (1968) 125.
J. J. Podesta andA. J. Arvia,Electrochem. Ada. 10 (1965) 159.
M. Schorr andJ. Yahalom,Corros. Sci. 12 (1972) 867.
R. M. Hudson, T. J. Buther andC. J. Warning,ibid. 17 (1977) 571.
Dhirendra, G. N. Pandey andB. Sanyal,Corr. Prevention Control 27(3) (1980) 13.
I. N. Putilova, S. A. Balezin andV. P. Barannik, “Metallic Corrosion Inhibitors” (Pergamon Press, Oxford, 1960) p. 27.
W. Machu,Korr. M. Metalloschutz 14 (1938) 324.
O. L. Riggs Jr andRay M. Hurd,Corrosion NACE 23 (1967) 252.
I. P. Anoschenko, in “Proceedings of the 3rd International Congress on Metallurgical Corrosion”, Tokyo 1966 (MIR, Moscow, 1969) p. 209.
K. E. Heusler andG. H. Cartledge,J. Electrochem. Soc. 108 (1961) 732.
L. Cavallara andL. Felloni,Electrochem. Ada 9 (1964) 485.
E. Mor, V. Scotto andC. Wrubl,Br. Corros. J. 7 (1972) 276.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sanad, S.H., Ismail, A.A. & Mahmoud, N.A. Inhibition effect of potassium iodide on corrosion of stainless steel in hydrochloric acid solution. J Mater Sci 27, 5706–5712 (1992). https://doi.org/10.1007/BF01119726
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01119726