Summary
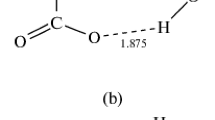
The complexes formed by simple association of the nickel atom with water and methanol, and those formed by insertion of nickel into O-H and O-C bonds, have been studied by the Linear Combination of Gaussian-type orbitalsdensity functional (LCGTO-DF) method. The binding energies calculated for theassociation complexes are 7 kcal/mol for both Ni(H2O) and Ni(CH3OH). These association complexes have equilibrium geometries characterized by a tetrahedral arrangement of bonds and lone pair around the oxygen atom. Theinsertion complexes HNiOH and HNiOCH3 are more stable than the association complexes by 14 and 18 kcal/mol, respectively. The H3CNiOH insertion complex is still more stable, by an additional 14 kcal/mol. In all five complexes, the lowest singlet and triplet states are nearly degenerate. Comparison of calculated frequencies with infrared spectra of matrix isolated species [MA Park (1988) PhD thesis, Rice University] indicates that only triplet states are seen in the experiment.
Similar content being viewed by others
References
Siegbahn PEM, Blomberg MRA (1993) In: van Leeuwen PWNM, van Lenthe JH, Morokuma K (eds) Theoretical aspects of homogeneous catalysis, applications of ab initio molecular orbital theory. Kluwer, Dordrecht
Fournier R (1993) J Chem Phys 98:8041
Bauschlicher CW Jr (1994) J Chem Phys 100:1215
Kasai PH, Jones PM (1985) J Am Chem Soc 107:813
Chenier JHB, Hampson CA, Howard JA, Mile B (1989) J Phys Chem 93:114
Castro M, Salahub DR, Fournier R (1994) J Chem Phys 100:8233
Villalta PW, Fenn PT, Leopold DG (1994) Proc SPIE, vol 2124: 325; and Leopold DG, private communication
Andzelm J, Wimmer E (1992) J Chem Phys 96:1280
Perdew JP, Chevary JA, Vosko SH, Jackson KA, Pederson MR, Singh DJ, Fiolhais C (1992) Phys Rev B46:6671
Becke AD (1992) J Chem Phys 96:2155; J Chem Phys (1992) 97:9173; J Chem Phys (1993) 98:5648
Deng L, Ziegler T, Fan L (1993) J Chem Phys 99:3823
Zhu T, Lee C, Yang W (1993) J Chem Phys 98:4814
Johnson BG, Gill PMW, Pople JA (1993) J Chem Phys 98:5612
Dickson RM, Becke AD (1993) J Chem Phys 99:3898
Malkin VG, Malkina OL, Salahub DR (1993) Chem Phys Lett 204:80; Chem Phys Lett 204:87
Colwell SM, Murray CW, Handy NC, Amos RD (1993) Chem Phys Lett 210:261
Eriksson L, Malkina OL, Malkin VG, Salahub DR (1994) J Chem Phys 100:5066
Verluis L, Ziegler T (1988) J Chem Phys 88:322; Fournier R, Andzelm J, Salahub DR (1989) J Chem Phys 90:6371; Dunlap BI, Andzelm J, Mintmire JW (1990) Phys Rev A 42:6354; Delley B (1991) J Chem Phys 94:7245
Berces A, Ziegler T (1993) J Chem Phys 98:4793
Fan L, Verluis L, Ziegler T, Baerends EJ, Ravenek W (1988) Int J Quantum Chem S22: 173
Fan L, Ziegler T (1992) J Phys Chem 96:6937
Pápai I, St-Amant A, Ushio J, Salahub DR (1990) Int J Quantum Chem Symp 24:29
Pápai I, Mink J, Fournier R, Salahub DR (1993) J Phys Chem 97:9986
Mitchell SA, Blitz MA, Fournier R (1994) Can J Chem 72:587
Brown CE, Mitchell SA, Hackett PA (1992) Chem Phys Lett 191:175
Mitchell SA (1992) In: Fontijn A (ed) Gas-phase metal reactions. Elsevier, Amsterdam, pp 227–252
Mitchell SA, Blitz MA, Siegbahn PEM, Svensson M (1994) J Chem Phys 100:423
Fournier R (1993) J Chem Phys 99:1801
Fournier R (1994) Int J Quantum Chem 52:973
Park MA (1988) PhD thesis, Rice University
Park MA, Hauge RH, Margrave JL (1988) High Temp Sci 25:1
Salahub DR, Fournier R, Mlynarski P, Papai I, St-Amant A, Ushio J (1991) In: Proc Ohio supercomputer center workshop on the theory and applications of density functional theory to chemistry. Labanowsky J, Andzelm J (eds) Springer, New York
St-Amant A, Salahub DR (1990) Chem Phys Lett 169:387; St-Amant A (1992) Thèse de doctorat, Université de Montréal
Vosko SH, Wilk L, Nusair M (1980) Can J Phys 58:1200
Becke AD (1988) Phys Rev A38:3098
Perdew JP (1986) Phys Rev B33:8822
Blomberg MRA, Brandemark UB, Siegbahn PEM (1986) Chem Phys Lett 126:317
Becke AD (1988) J Chem Phys 88:2547
Jones RS, Mintmire JW, Dunlap BI (1988) Int J Quantum Chem Symp 22:77
Jansen HB, Ros P (1969); Chem Phys Lett 3:140; Boys SF, Bernardi F (1970) Mol Phys 19:553; Gutowski M, Chałasiński G (1993) J Chem Phys 98:5540; Davidson ER, Chakravorty SJ (1994) Chem Phys Lett 217:48 (1994)
Goursot A, Pápai I, Daul CA (1994) Int. J Quantum Chem 52:799
Shimanouchi T (1977) Tables of molecular vibrational frequencies I, Publication NSRDS-NBS 39, National Bureau of Standards, Washington DC; Shimanouchi T, Tables of molecular vibrational frequencies II, J Phys Chem Ref Data 6:993
Blomberg MRA, Brandemark UB, Siegbahn PEM, Mathisen KB, Karlström G (1985) J Phys Chem 89:2171
Bauschlicher CW Jr (1986) J Chem Phys 84:260
Bauschlicher CW Jr (1987) Chem Phys Lett 142:71
Sunderlin LS, Wang D, Squires RR (1992) J Am Chem Soc 114:2788
We use six CGd orbitals:xx, xy, xz, yz, andzz. The population of these orbitals in a closed d shell are 1 1/3, 2, 2, 1 1/3, 2, and 1 1/3, respectively
Siegbahn PEM, Blomberg MRA, Svensson M (1993) J Phys Chem 97:2564
Bauschlicher CW Jr, Langhoff SR, Partridge H (1986) J Chem Phys 84:901; Bauschlicher CW Jr (1986) Int J Quantum Chem S20:563
Ball DW, Hauge RH, Margrave JL (1988) High Temp Sci 25:95
Park et al. did assign some bands (3680, 1785 and 631 cm−1) to HNiOH(H2O) but did not consider the possibility of a second isomer of HNiOH(H2O)
In general, the barrier to metal insertion in bonds increase in that order: H-H, C-H, C-C [1]. This has been rationalized from the directional nature of C-X bonds as follows (see Ref. [1] and references therein). The necessary distortion of the bonds around the carbon atom away from tetrahedral arrangement, which is needed to reach the transition state, is very unfavourable energetically. This energy cost is lower for C-H bonds than for C-C bonds — and is still lower for the H-H bond — because a H atom is spherical and can bind both to the metal and the other R group more effectively than can a methyl group. Using that same idea, we expect the barrier to insertion in a O-C bond to be larger than that for insertion in a O-H bond.
Fan L, Ziegler T (1990) J Chem Phys 92:3645
Burghgraef H, Jansen APJ, van Santen RA (1993) J Chem Phys 98:8810
Abashkin Y, Russo N (1994) J Chem Phys 100:4477
Stanton RV, Merz KM Jr (1994) J Chem Phys 100:434
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fournier, R. Association and insertion complexes of nickel with water and methanol studied using Kohn-Sham theory. Theoret. Chim. Acta 91, 129–146 (1995). https://doi.org/10.1007/BF01114981
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01114981