Abstract
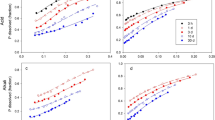
The effect of phosphate (P), calcium (Ca), and pH on the dissolution of Sechura phosphate rock (SPR) in a Typic Dystrochrept was investigated in an incubation study over 90 days. Increasing the P status of the soil had little effect on either the rate or extent of dissolution of SPR, as measured by a single extraction with 0.5M NaOH, or on the amount of plant-available P in the soil as measured by the Bray procedure. This suggests that a reactive PR such as SPR could be effective as a maintenance P fertilizer on soils of medium and high P status.
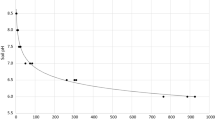
The dissolution of SPR at pH 6.4 was only 4% of total P added, compared to 33.6% of total P added at pH 5.2. Addition of Ca as CaCl2, at the same rate of Ca as that added in Ca(OH)2 to raise soil pH from 5.2 to 6.4, reduced the dissolution of SPR to 11% of total P added. The large effect of Ca on the dissolution of SPR contrasts with the small effect of soil P status and probably reflects the fact that the concentration of Ca in the soil solution is generally several orders of magnitude higher than that of P. The results obtained suggest that pH, per se, is of less importance in the dissolution and subsequent plant availability of P from phosphate rock in soil than previously thought.
Similar content being viewed by others
References
Association of Official Analytical Chemists (1960) ‘Official Methods of Analysis’, 9th ed, Sec 2.031b
Barnes JS and Kamprath EJ (1975) Availability of North Carolina rock phosphate applied to soils. North Carolina Agric Exp Stat Techn Bull No. 229
Bray RH and Kurtz LT (1945) Determination of total organic and available forms of phosphorus in soil. Soil Sci 59:39–45
Bremner JM and Jenkinson DS (1960) Determination of organic carbon in soil. I. Oxidation by dichromate of organic matter in soil and plant materials. J Soil Sci 11:394–402
Chang SC and Jackson ML (1957) Fractionation of soil phosphorus. Soil Sci 84:133–144
Chien SH, Leon LA and Tejeda HR (1980) Dissolution of North Carolina phosphate rock in acid Colombian soils as related to soil properties. Soil Sci Soc Amer J 44:1267–1271
Chu CR, Moschler WW and Thomas GW (1962) Rock phosphate transformations in acid soils. Soil Sci Soc Amer Proc 26:476–478
Ellis RJ, Quader RA and Truog E (1955) Rock phosphate availability as influenced by soil pH. Soil Sci Soc Amer Proc 19:484–487
Ensminger LE, Pearson RW and Armiger WH (1967) Effectiveness of rock phosphate as a source of phosphorus for plants. USDA ARS, pp 41–125. Washington, DC
Joos LL and Black CA (1950) Availability of phosphate rock as affected by particle size and contact with bentonite and soil of different pH values. Soil Sci Soc Amer Proc 15:69–75
Juo ASR and Kang BT (1978) Availability and transformation of rock phosphates in three forest soils from southern Nigeria. Commun Soil Sci Plant Anal 9:493–505
Khasawneh FE and Doll EC (1978) The use of phosphate rock for direct application to soils. Adv Agron 30:159–206
Mackay AD, Gregg PEH and Syers JK (1980) A preliminary evaluation of Chatham Rise phosphorite as a direct-use phosphatic fertilizer. NZ J Agric Res 23:441–449
Mackay AD, Gregg PEH and Syers JK (1984) Field evaluation of Chatham Rise phosphorite as a phosphatic fertilizer for pasture. NZ J Agric Res 27:65–82
Mackay AD, Syers JK, Tillman RW and Gregg PEH (1984) A comparison of 3 soil-testing procedures for estimating the plant availability of phosphorus in soils receiving either superphosphate or phosphate rock. NZ J Agric Res 27:231–245
Mackay AD, Syers JK, Tillman RW and Gregg PEH (1986) A simple model to describe the dissolution of phosphate rock in soils. Soil Sci Soc Amer J 50: (In press)
O'Connor PW and Syers JK (1975) Comparison of methods for the determination of total phosphorus in waters containing particulate material. J Environ Qual 4:347–350
Rajan SSS (1981) ‘Biosuper’ and phosphate rock as phosphate fertilizers. Recent studies at Ruakura. Proceedings Technical Workshop on Potential of Phosphate Rock as a Direct Application fertilizer in New Zealand. Massey University, Palmerston North. Occasional Report No. 3, pp. 32–39
Ryden JC and Syers JK (1975) Charge relationships of phosphate sorption. Nature 255:51–53
Saunders WMB (1965) Phosphate retention by New Zealand soils and relationships to free sesquioxides, organic matter and other soil properties. NZ J Agric Res 8:30–57
Shinde BN, Sarangamath PA and Patnaik S (1978) Phosphorus transformations from rock phosphates in acid soils and measures for increasing their efficiency for growing rice (Oryza sativa L.) Plant Soil 49:449–459
Smillie GW and Syers JK (1972) Calcium fluoride formation during extraction of calcareous soils with fluoride. II. Implications to the Bray P-1 Test. Soil Sci Soc Amer Proc 36:25–30
Syers JK and Gregg PEH (1981) Potential of Phosphate Rock as a Direct Application fertilizer in New Zealand. 168 pp. Proceedings of a Technical Workshop Massey University, Palmerston North. Occasional Report No. 3
Syers JK and Mackay AD (1986) Reaction of Sechura phosphate rock and single superphosphate in soil. Soil Sci Soc Amer J 50: (In press)
The Fertilisers Regulations (1969) Document 1969–88. Government printer, Wellington, 34 pp
Van der Paauw F (1965) Factors controlling the efficiency of rock phosphates for potatoes and rye on humic sandy soils. Plant Soil 22:81–98
Williams JDH, Syers JK and Walker TW (1967) Fractionation of soil inorganic phosphate by a modification of Chang and Jackson's procedure. Soil Sci Soc Amer Proc 31:736–739
Wilson MA and Ellis BG (1984) Influence of calcium solution activity and surface area on the solubility of selected rock phosphates. Soil Sci 138:354–359
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mackay, A., Syers, J. Effect of phosphate, calcium, and pH on the dissolution of a phosphate rock in soil. Fertilizer Research 10, 175–184 (1986). https://doi.org/10.1007/BF01074371
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01074371