Summary
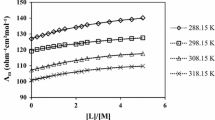
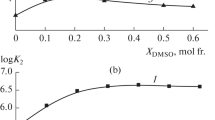
The kinetics of the solvolysis of thetrans-[Coen2Cl2]+ cation (en = 1,2-diaminoethane) have been investigated over a range of temperatures in water with added ethanonitrile which has much less effect on the solvent structure than the acohols already used as co-solvents with water for this solvolysis. However, the non-linear relationship obtained for the variation of log (rate constant) with the reciprocal of the dielectric constant at constant temperature shows that the effect of changing solvent structure is still important, although, as expected from the absence of pronounced extrema in the variation with composition in water + ethanonitrile of physical properties which are influenced by such changes in solvent structure, the enthalpy and entropy of activation for the solvolysis vary smoothly with composition. The application of a free energy cycle shows that changes in solvent structure affect the pentacoordinated cobalt(III) ion in the transition state more than the hexacoordinated cobalt(III) ion in the initial state.
Similar content being viewed by others
References
G. S. Groves and C. F. Wells,J. Chem. Soc., Faraday Trans. I, 78 619 (1982).
G. S. Groves and C. F. Wells,J. Chem. Soc., Faraday Trans. I, 81, 2475 (1985).
H. S. Frank and M. W. Evans,J. Chem. Phys., 13, 507 (1945); H. S. Frank and W.-Y. Wen,Discussion Faraday Soc., 24, 133 (1957); G. Némethy and H. A. Sheraga,J. Chem. Phys., 36, 3382, 3401 (1962); W. Laiden and G. Némethy,J. Phys. Chem., 74, 3501 (1970).
G. Wada and S. Umeda,Bull. Chem. Soc. Jpn., 35, 646 (1962).
K. Nakanishi,Bull. Chem. Soc. Jpn., 33, 793 (1960).
J. Kenttämaa, E. Tommila and M. Martti,Ann. Acad. Scient. Fenn., No. 93 (1959).
M. J. Blandamer,Introduction to Chemical Ultrasonics, Academic Press, London, 1973, Ch. 11.
R. F. Lama and B. C.-Y. Lu,J. Chem. Eng. Data, 10, 216 (1965).
K. J. Laidler and H. Eyring,Ann. N. Y. Acad. Sci., 39, 303 (1940); S. Glasstone, K. J. Laidler and H. Eyring,The Theory of Rate Processes, McGraw-Hill, New York, 1941, Ch. 8; K. J. Laidler and P. Landskroener,Trans. Faraday Soc., 52, 200 (1956); K. J.Laidler,Suomen Kemistil, A, 33, 44 (1960); K. J. Laidler,Chemical Kinetics, McGraw-Hill, New York, 2nd Edit. 1965, Ch. 5.
E. A. Moelwyn-Hughes,Proc. Roy. Soc. London, Ser., A155, 308 (1936); Ibid., 157, 667 (1936),The Kinetics of Reactions in Solution, Oxford Univ. Press, London, 2nd Edit., 1947, Chs. 4, 5 and 7;Physical Chemistry, Pergamon Press, Oxford, 2nd Edit., 1961, Chs. 7–9, 24.
E. S. Amis,Kinetics of Chemical Change in Solution, Macmillan, New York, 1949, Chs. 5 and 9;Solvent Effects on Reaction Rates and Mechanism, Academic Press, New York, 1966, Chs. 1–3;Solvent Effects on Chemical Phenomena, Academic Press, New York, 1973, Vol. 1, Ch. 5.
C. Treiner, P. Tzias, M. Chemla and G. M. Poltovatskii,J. Chem. Soc., Faraday Trans. I, 72, 2007 (1976).
D. A. Armitage, M. J. Blandamer, M. J. Foster, N. J. Hidden, K. W. Morcom, M. C. R. Symons and M. J. Wootten,Trans. Faraday Soc., 64, 1193 (1968).
C. Moreau and G. Douhéret,Thermochim. Acta, 13, 385 (1975).
A.-L. Vierk,Z. Anorg. Chem., 261, 283 (1950).
J. Burgess,J. Chem. Soc. A, 2351 (1970).
R. G. Pearson, C. R. Boston and F. Basolo,J. Am. Chem. Soc., 75, 3089 (1953).
C. Moreau and G. Douhéret,J. Chem. Thermodynamics, 8, 403 (1976).
E. Grunwald and S. Winstein,J. Am. Chem. Soc., 70, 846 (1948); S. Winstein, E. Grunwald and H. W. Jones,J. Am. Chem. Soc., 73, 2700 (1951).
R. E. Robertson and S. E. Sugamori,Can. J. Chem., 50, 1353 (1972).
R. E. Robertson and S. E. Sugamori,J. Am. Chem. Soc. 91, 7254 (1969).
F. Akhtar and R. A. Begum,J. Bangladesh Acad. Sci., 2, 9 (1978).
W. E. Jones, L. R. Carey and J. W. Swaddle,Can. J. Chem., 56, 2739 (1972); G. A. Lawrance,Inorg. Chim. Acta, 45, L 275 (1980); G. A. Lawrance and S. Suvachittanont,Austr. J. Chem., 33, 277 (1980); D. A. Palmer and H. Keim,Inorg. Chem., 16, 3139 (1977);Coord. Chem. Rev., 36, 89 (1981); G. Daffner, D. A. Palmer and H. Kelm,Inorg. Chim. Acta, 61, 57 (1982).
W. G. Jackson and A. M. Sargeson,Inorg. Chem., 17, 1348 (1978); W. G. Jackson and C. M. Begbie,Inorg. Chim. Acta, 60, 115 (1982).
C. F. Wells,J. Chem. Soc., Faraday Trans I, 73, 1851 (1977).
G. S. Groves,Ph. D. Thesis, Univ. of Birmingham, 1983).
G. S. Groves and C. F. Wells,J. Chem. Soc., Faraday Trans. I, 81, (1985).
C. F. Wells,Austral. J. Chem., 36, 1739 (1983).
C. F. Wells,J. Chem. Soc., Faraday Trans. I, 69, 984 (1973);70, 694 (1974);71, 1868 (1975);72, 601 (1976);74, 636,1569 (1978);77, 1515 (1981);80, 2445 (1984);Adv. Chem. Ser., 177, 53 (1979);Thermochim. Acta, 53, 67 (1982); G. S. Groves and C. F. Wells,J. Chem. Soc., Faraday Trans. I, in press; G. S. Groves, I. M. Sidahmed and C. F. Wells, unpublished results.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Groves, G.S., Wells, C.F. Kinetics of the solvolysis of thetrans-dichlorobis(1,2-diaminoethane) cobalt(III) in water + ethanonitrile mixtures. Transition Met Chem 11, 20–23 (1986). https://doi.org/10.1007/BF01064495
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01064495