Abstract
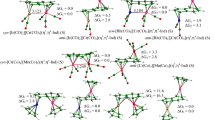
The NDDO method has been extended to include elements withd-orbitals. A parametrization for the first-row transition metals is given, which has been worked out to describe structural properties of transition metal complexes. Applications of the method to the Jahn-Teller distortions of tetrachloro complexes are presented. The NDDO results are compared with those from CNDO and INDO calculations. It turns out that the NDDO method seems to be the most sensitive among the ZDO procedures.
Similar content being viewed by others
References
Pople, J. A., Santry, D. P., Segal, G. A.: J. Chem. Phys.43, S129 (1965)
Baetzold, R. C.: J. Chem. Phys.55, 4355 (1971)
Clack, D. W., Hush, N. S., Yandle, J. R.: J. Chem. Phys.57, 3503 (1972)
Blyholder, G.: J. Phys. Chem.79, 756 (1975)
Serafini, A., Savariault, J. M., Cassoux, P., Labarre, J. F.: Theoret. Chim. Acta (Berl.)36, 241 (1975); Serafini, A., Pelissier, M., Savariault, J. M., Cassoux, P., Labarre, J. F.: Theoret. Chim. Acta (Berl.)39, 229 (1975)
Tatsumi, K., Fueno, T.: Bull. Chem. Soc. Japan49, 929 (1976)
Freund, H. J., Hohlneicher, G.: Theoret. Chim. Acta (Berl.)51, 145 (1979)
Bacon, A. D., Zerner, M. C.: Theoret. Chim. Acta (Berl.)53, 21 (1979)
Rajzmann, M., Francois, P., Charles, P.: J. Chim. Phys.76, 328 (1979)
Golebiewski, A., Witko, M.: Acta Phys. Polon57, 585 (1980)
Kai, E., Nishimoto, K.: Int. J. Quant. Chem.18, 403 (1980)
Bhattacharyya, S. P.: Proc. Indian Acad. Sci.89, 183 (1980)
Böhm, M. C., Gleiter, R.: Theoret. Chim. Acta (Berl.)59, 127 (1981); 153 (1981)
Baba-Ahmed, A., Gayoso, J.: Theoret. Chim. Acta (Berl.)62, 507 (1983)
Lüthi, H. P., Ammeter, J. H., Almlöf, J., Faegri, K.: J. Chem. Phys.77, 2002 (1982)
Bachelet, G. B., Hamann, D. R., Schlüter, M.: Phys. Rev.B26, 4199 (1982)
Sustmann, R., Williams, J. E., Dewar, M. J. S., Allen, L. C., Schleyer, P. v. R.: J. Am. Chem. Soc.91, 5350 (1969)
Birner, P., Köhler, H. J., Weiss, C.: Chem. Phys. Letters27, 347 (1974)
Thiel, W.: Theoret. Chim. Acta (Berl.)59, 191 (1981)
Fletcher, R.: Atomic energy research establishment report AERE-R 7125 (1972)
Roothaan, C. C. J.: J. Chem. Phys.19, 1445 (1951)
Birner, P.: Programmsystem NDDO80
Harris, F. E.: J. Chem. Phys.51, 4770 (1969)
Goeppert-Mayer, M., Sklar, A. L.: J. Chem. Phys.6, 645 (1938)
Ruedenberg, K.: J. Chem. Phys.19, 1433 (1951)
Pople, J. A., Beveridge, D. L.: Approximate molecular orbital theory, New York: McGraw-Hill Co. 1970
Tables of interatomic distances and configurations in molecules and ions, Supplement, Sutton, L. E., Ed. London: The Chem. Soc. 1965
Morino, Y., Uehara, H.: J. Chem. Phys.45, 4543 (1966)
Basch, H., Viste, A., Gray, H. B.: J. Chem. Phys.44, 10 (1966)
Figgis, B. N., Gerlock, M., Mason, R.: Acta Cryst.17, 506 (1964)
Weisner, J. R., Srivastava, R. C., Kennard, C. H. L., DiVaira, M., Lingafelter, E. C.: Acta Cryst.23, 565 (1967)
Morosin, B., Lingafelter, E. C.: J. Chem. Phys.65, 50 (1961)
Jahn, H. A., Teller, E.: Proc. Roy. Soc. (London), A161 (1937)
Gill, N. S.: J. Chem. Soc. 3512 (1961)
Pelikan, P., Liska, M.: Collect. Czech. Chem. Commun.47, 1556 (1982)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nieke, C., Reinhold, J. Application of the NDDO method to transition metal compounds. Theoret. Chim. Acta 65, 99–108 (1984). https://doi.org/10.1007/BF01064206
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01064206