Abstract
The livers of 15 rabbits were perfused in situwith prednisone (PO) or prednisolone (POH) over a wide range of steady state concentrations, resulting in multiple experimental measurements per organ. Linearity of extraction, an apparent lack of oxidative conversion, and marked preference for the reduction of PO to POH was observed. Predictions of hepatic tissue concentrations were made using both the well-stirred and parallel-tube model approximations. Giucocorticoid disposition across the liver was described by a series of differential equations. Discrimination between the two models was accomplished by examining the effects of changes inflow rate upon the availability of the highly extracted drug PO. The well-stirred model very closely predicted the observed changes in availability of PO, whereas the parallel-tube model provided poor predictions. The intrinsic clearances of interconversion and elimination of PO and POH were subsequently calculated by population analysis using NONMEM. This method assumed the well-stirred model and resulted in intrinsic clearance estimates of 26 ml/min for the elimination of POH, 157 ml/min for reductive conversion of PO to POH, and 205 ml/min for the irreversible elimination of PO. A mechanism of intrahepatic disposition of these glucocorticoids was proposed using well-stirred model predictions of hepatic drug concentrations, the perfusion rate limitation to drug transport, and the assumption of no oxidative interconversion of POH to PO. In this case, the capacity for reduction of PO to POH approaches the elimination clearance of PO and the elimination of PO is about 13 times greater than the elimination clearance of POH.
Similar content being viewed by others
Abbreviations
- PO, POH:
-
As superscripts refer to the unbound concentrations of prednisone and prednisolone, respectively
- C H :
-
Unbound concentration of drug within the hepatocyte, as predicted by either the well-stirred or parallel-tube model
- C in :
-
Unbound concentration of drug in perfusate entering the liver, as measured at the entrance sampling port
- C out :
-
Unbound concentration of drug in perfusate exiting the liver, as measured at the exit sampling port
- dC/dt :
-
Differential expression describing the time-dependent disposition of drug concentration across the liver
- CL int :
-
Intrinsic hepatic clearance of drug
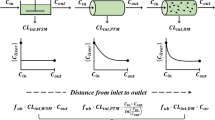
- CL 10,CL 12,CL 20,CL 21 :
-
Intrinsic clearances, as defined in Fig. 1
- E :
-
Extraction ratio of drug, defined as (C in-C out)/C in
- 1−E :
-
Availability of drug
- E ws :
-
Hepatic extraction of drug, assuming the well-stirred model assumption
- E pt :
-
Hepatic extraction of drug, assuming the parallel-tube model assumption
- E exp :
-
Extraction of drug, obtained experimentally
- E idel :
-
Extraction of drug at the ideal flow rate (at 20 mm Hg perfusion pressure)
- fu :
-
Unbound fraction of drug in perfusate
- NONMEM:
-
Nonlinear Mixed Effects Modeling program
- Obj. Funct.:
-
Objective function
- PT:
-
Parallel-tube model of organ disposition
- G H :
-
Flow rate of perfusate (ml/min)
- V H :
-
Volume of liver (ml), assuming a density of 1 g/ml
- WS:
-
Well-stirred model of organ disposition
- ɛ :
-
Residual intraliver error
- η j :
-
Randomly distributed interliver errors of clearance terms with a mean of zero
- ϕ :
-
Population mean estimate of clearance
References
M. E. Pickup, J. R. Lowe, P. A. Leatham, V. M. Rhind, V. Wright, and W. W. Downie. Dose-dependent pharmacokinetics of prednisone and prednisolone.Eur. J. Clin. Pharmacol. 12:213–219 (1977).
J. C. K. Loo, I. J. McGilveray, N. Jordan, J. Moffat, and R. Brien. Dose-dependent pharmacokinetics of prednisone and prednisolone in man.J. Pharm. Pharmacol. 30:736 (1978).
J. Q. Rose, A. M. Yurchak, and W. J. Jusko. Dose-dependent pharmacokinetics of prednisone and prednisolone in man.J. Pharmacokin. Biopharm. 9:389–417 (1981).
U. F. Legler, F. J. Frey, and L. Z. Benet. Prednisolone clearance at steady state in man.J. Clin. Endocrinol. Metab. 55:762–767 (1982).
F. J. Frey, B. M. Frey, A. Greither, and L. Z. Benet. Prednisolone clearance at steady state in dogs.J. Pharmacol. Exp. Ther. 215:9 (1981).
M. Hammarlund-Udenaes and L. Z. Benet. Prednisone and prednisolone interconversion in the rabbit utilizing unbound concentrations.Biopharm. Drug Dispos. 12:467–477 (1991).
L. T. Samuels and C. D. West. The intermediary metabolism of the non-benzenoid steroid hormones.Vitam. Harm. 10:251–295 (1952).
S. Miyabo, S. Kishida, and T. Hisada. Metabolism and conjugation of cortisol by various dog tissuesin vitro.J. Steroid Biochem. 4:567–576 (1973).
M. J. O'Hare. Corticosterone metabolism by rat liver adrenocortical cell suspension and monolayer cultures: the effect of damage on 11-dehydrogenase activity.J. Endocrinol. 59:141–150 (1973).
A. L. Bernal, A. B. M. Anderson, and A. C. Turnbull. Cortisol: cortisone interconversion by human decidua in relation to parturition: effect of tissue manipulation on 11/3-hydroxystereoid dehydrogenase activity.J. Endocrinol. 93:141–149 (1982).
V. G. Hale and L. Z. Benet. Prednisolone and prednisone exhibit linear extraction in the perfused rabbit liver.Drug Metab. Dispos. 19:87–93 (1991).
F. J. Frey, B. M. Frey, and L. Z. Benet. Liquid Chromatographic measurement of endogenous and exogenous glucocorticoids in plasma.Clin. Chem. 25:1944–1947 (1979).
L. E. Gustavson and L. Z. Benet. The macromolecular binding of prednisone in the plasma of healthy volunteers including pregnant women and oral contraceptive users.J. Pharmacokin. Biopharm. 13:561–569 (1985).
K. S. Pang and M. Rowland. Hepatic clearance of drugs. II. Experimental evidence for acceptance of the well stirred model over the parallel tube model using lidocaine in the perfused rat liverin situ preparation.J. Pharmacokin. Biopharm. 5:655–680 (1977).
K. S. Pang and M. Rowland. Hepatic clearance of drugs. I. Theoretical considerations of a “well stirred” model and a “parallel tube” model. Influence of hepatic blood flow, plasma blood cell binding, and the hepatocellular enzymatic activity on hepatic drug clearance.J. Pharmacokin. Biopharm. 5:625–653 (1977).
S. L. Beal and L. B. Sheiner. Methodology of population pharmacokinetics. In E. R. Garrett and J. L. Hirtz (eds.),Drug Fate and Metabolism, Marcel Dekker, New York, 1985, pp. 135–183.
S. L. Beal, L. B. Sheiner, and A. Boeckmann: NONMEM Users Guides, Technical Report of the Division of Clinical Pharmacology, University of California San Francisco, 1985.
A. B. Ahmad, P. N. Bennett, and M. Rowland. Models of hepatic drug clearance: discrimination between the ‘well stirred’ and ‘parallel tube’ models.J. Pharm. Pharmacol. 35:219–224 (1983).
S. Keiding and E. Chiarantini. Effect of sinusoidal perfusion on galactose elimination in the perfused rat liver.J. Pharmacol. Exp. Ther. 205:465–470 (1978).
S. Keiding and E. Steiness. Flow dependence of propranolol elimination in the perfused rat liver.J. Pharmacol. Exp. Ther. 230:474–477 (1984).
R. W. Brauer. Liver circulation and function.Physiol. Rev. 43:115–213 (1963).
E. L. Forker and B. Luxon. Hepatic transport kinetics and plasma disappearance curves: distributed modeling versus conventional approach.Am. J. Physiol. 235:E648-E660 (1978).
M. S. Roberts and M. Rowland. A dispersion model of hepatic elimination: I. Formulation of the model and bolus considerations.J. Pharmacokin. Biopharm. 14:227–260 (1986).
B. E. P. Murphy. Ontogeny of cortisol-cortisone interconversion in human tissues: a role for cortisone in human fetal development.J. Steroid Biochem. 14:811–817 (1981).
D. R. Koerner. 11Β-Hydroxysteroid dehydrogenase of lung and testis.Endocrinology 79:935–938 (1966).
C. Monder and C. H. L. Shackleton. 11Β-Hydroxysteroid dehydrogenase: Fact or fancy?Steroids 44:383–417 (1984).
J. Kawamura, T. Hayakawa, and T. Tanimoto. Steroid-protein interactions: substrate specificity of 20Β-hydroxysteroid dehydrogenase.Chem. Pharm. Bull. 28:437–446 (1980).
B. Hurlock and P. Talalay. Microsomal 3α- and 11Β-hydroxysteroid dehydrogenase.Arch. Biochem. Biophys. 80:468–470 (1959).
Author information
Authors and Affiliations
Additional information
Supported in part by NIH grant GM 26691. During the course of the experimental studies performed at the Department of Pharmacy, University of California, San Francisco, V. G. Hale was supported as a predoctoral fellow by NIH Pharmacological Sciences Training Grant GM 07175 and as a fellow of the American Foundation for Pharmaceutical Education.
Rights and permissions
About this article
Cite this article
Hale, V.G., Aizawa, K., Sheiner, L.B. et al. Disposition of prednisone and prednisolone in the perfused rabbit liver: Modeling hepatic metabolic processes. Journal of Pharmacokinetics and Biopharmaceutics 19, 597–614 (1991). https://doi.org/10.1007/BF01062965
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01062965