Abstract
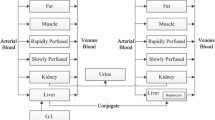
Various pharmacokinetic parameters—disposition half-life, t 1/2,z, metabolic clearance CLm, volume of distribution V, intrinsic clearance of unbound drug CLut, and unbound volume of distribution of tissues (distributive tissue volume / fraction of drug in tissue unbound, VT/fuT—are compared in rat and human for nine weakly acidic drugs, phenytoin, hexobarbital, pentobarbital, phenylbutazone, warfarin, tolbutamide, valproate, phenobarbital, and amobarbital, and six weakly basic drugs, quinidine, chlorpromazine, propranolol, pentazocin, antipyrine, and diazepam. With regard to all parameters, statistically significant correlations are obtained when parameters are plotted on a log-log plot. Correlation coefficients between the intrinsic parameters (CLuint or VT/fuT) were higher than those between the hybrid parameters (t1/2,z, CLm, or V). In general, these drugs were metabolized ten times more rapidly in rat than in human. With regard to the tissue distribution of these drugs, there was little difference between rat and human. Predictions of CLm, V, and t1/2, in humans using rat data were successful for most drugs, with a few marked exceptions.
Similar content being viewed by others
References
L. B. Mellett. Comparative drug metabolism.Prog. Drug Res,13:136–169 (1969).
R. L. Dedrick, K. B. Bischoff, and D. S. Zaharko. Interspecies correlation of plasma concentration history of methotrexate.Cancer Chemother. Part 1 54:95–101 (1970).
R. L. Dedrick. Animal scale up.J. Pharmacokin. Biopharm. 1:435–461 (1973).
U. Klotz, K. H. Antonin, and P. R. Bieck. Pharmacokinetics and plasma binding of diazepam in man, dog, rabbit, guinea pig, and rat.J. Pharmacol. Exp. Ther. 199:67–73 (1976).
C. H. Walker. Species differences in microsomal monoxygenase activity and their relationship to biological half-lives.Drug Metab. Rev. 7:295–323 (1978).
C. H. Walker. Species variations in some hepatic microsomal enzymes that metabolize xenobiotics. In: J. W. Bridges and L. F. Chasseaud (eds.),Progress in Drug Metabolism, Vol. 5, Wiley, London, 1980, Chapter 2, pp. 113–164.
R. L. Dedrick, D. D. Forester, J. N. Cannon, S. M. E. Dareen, and L. B. Mellett. Pharmacokinetics of 1-β-spd-arabino-furanosylcytosine (Ara-C) deamination in several species.Biochem. Pharmacol. 22:2405–2417 (1973).
H. Boxenbaum. Interspecies variation in liver weight, hepatic blood flow, and antipyrine intrinsic clearance Extrapolation of data to benzodiazepines and phenytoin.J. Pharmacokin. Biopharm. 8:165–176 (1980).
H. Boxenbaum. Interspecies scaling, allometry, physiological time, and the ground plan of pharmacokinetics.J. Pharmacokin. Biopharm. 10:201–227 (1982).
H. Boxenbaum. Comparative pharmacokinetics of benzodiazepines in dog and man.J. Pharmacokin. Biopharm. 10:411–426 (1982).
Y. Sawada, M. Hanano, Y. Sugiyama, and T. Iga. Prediction of the disposition ofβ-lactum antibiotics in human from pharmacokinetic parameters in animals.J. Pharmacokin. Biopharm. 12:241–261 (1984).
L. Z. Benet and L. B. Sheiner. Design and optimization of dosage regimens; Pharmacokinetic data. In L. S. Goodman and A. Gilman (eds.),The Pharmacological Basis of Therapeutics, 6th ed. Macmillan, New York, 1980, pp. 699, 1737.
R. G. Dickison, R. C. Harland, A. M. Ilias, R. M. Rodgers, S. N. Kaufman, R. K. Lynn, and N. Gerber. Disposition of valproic acid in the rat: Dose-dependent metabolism, distribution, enterohepatic recirculation and choleretic effect.J. Pharmacol. Exp. Ther. 211:583–595 (1979).
Y. Tanimura. Pharmacokinetics of Phenylbutazone and Its Metabolites in Rats. Ph.D. thesis, University of Tokyo (1979).
T. Inaba and T. Umeda. Biliary excretion of diphenylhydantoin in the rat.Drug Metab. Dispos. 3:69–73 (1975).
T. Inaba, E. Tsutsumi, W. A. Mahon, and W. Kalow. Biliary excretion of diazepam in the rat.Drug Metab. Dispos. 2:429–432 (1974).
F. Ichimura, K. Yokogawa, T. Yamana, A. Tsuji, and Y. Mizukami. Physiological pharmacokinetic model for pentazoxine. I. Tissue distribution and elimination in the rat.Int. J. Pharm. 15:321–333 (1983).
K. S. Pang and M. Rowland. Hepatic clearance of drugs. I. Theoretical considerations of a “well-stirred” model and a “parallel tube” model. Influence of hepatic blood flow, plasma and blood cell binding, and the hepatocellular enzyme activity on hepatic drug clearance.J. Pharmacokin. Biopharm. 5:625–653 (1977).
G. R. Wilkinson and D. G. Shand. A physiological approach to hepatic drug clearance.Clin. Pharmacol. Ther. 18:377–390 (1975).
O. Sugita, Y. Sawada, Y. Sugiyama, T. Iga, and M. Hanano. Effect of sulphaphenazole on tolbutamide distribution in rabbits. Analysis of interspecies difference in tissue distribution of tolbutamide.J. Pharm. Sci. 73:631–634 (1984).
J. W. Prothero. Scaling of blood parameters in mammals.Comp. Biochem. Physiol. 67A:649–657 (1980).
U. K. Hansen. Molecular aspect of ligand binding to serum albumin.Pharmacol. Rev. 33: 17–53 (1981).
K. M. Piafsky. Disease-induced changes in the plasma binding of basic drugs.Clin. Pharmacokin. 5:246–262 (1980).
M. K. Romach, K. M. Piafsky, J. G. Abel, V. Khouw, and E. M. Sellers. Methasone binding to orosomucoid (α 1-acid glycoprotein): Determinant of free fraction in plasma.Clin. Pharmacol. Ther. 29:211–217 (1981).
S. Glasson, R. Zili, P. D. Athis, J. P. Tillement, and J. R. Boissier. The distribution of bound propranolol between the different human serum proteins.Mol. Pharmacol. 17:187–191 (1980).
W. E. Muller and A. E. Stillbaluer. Characterization of a common binding site for basic drugs on humanα 1-acid glycoprotein (orosomucoid).Naunyn-Schmiedeberg's Arch. Pharmacol. 322:170–173 (1983).
J. J. Lima, H. Boudoulas, and M. Blanford. Concentration-dependence of disopyramide binding to plasma protein and its influence on kinetics and dynamics.J. Pharmacol. Exp. Ther. 219:741–747 (1981).
O. G. Nilsin. Serum albumin and lipoproteins as the quinidine binding molecules in normal human sera. Biochem.Pharmacol. 25:1007–1012 (1976).
A. Yacobi and G. Levy. Comparative pharmacokinetics of coumarin and anticoagulants XIV: Relationship between protein binding distribution and elimination kinetics of warfarin in rats.J. Pharm. Sci. 64:1660–1664 (1975).
A. Yacobi, J. A. Udall, and G. Levy. Serum protein binding as a determinant of warfarin body clearance and anticoagulant effect.Clin. Pharmacol. Ther. 19:552–558 (1976).
G. H. Evans, A. S. Nies, and D. G. Shand. The disposition of propanolol. III. Decreased half-life and volume of distribution as a result of plasma binding in man, monkey, dog and rat.J. Pharmacol. Exp. Ther. 186:114–122 (1973).
D. Fremstad, O. G. Nilsen, L. Storstein, J. Amlie, and S. Jacobsen. Pharmacokinetics of quinidine related to plasma protein binding in man.Eur. J. Clin. Pharmacol. 15:187–192 (1979).
W. A. Colburn and M. Gibaldi. Plasma protein binding and metabolic clearance of phenytoin in the rat.J. Pharmacol. Exp. Ther. 203:500–506 (1977).
Y. Igari, Y. Sugiyama, Y. Sawada, T. Iga, and M. Hanano. Prediction of diazepam disposition in the rat and man by a physiologically based pharmacokinetic model.J. Pharmacokin. Biopharm. 11: 577–593 (1983).
H. Harashima, Y. Sawada, Y. Sugiyama, T. Iga, and M. Hanano. Analysis of non-linear tissue distribution of quinidine in rats by physiologically based pharmacokinetics.J. Pharmacokin. Biopharm. 13:425–440 (1985).
T. Itoh, Y. Sawada, T. Iga, and M. Hanano. Unpublished data.
Y. Igari, Y. Sugiyama, S. Awazu, and M. Hanano. Comparative physiologically based pharmacokinetics of hexobartital, phenobarbital and thiopental in the rat.J. Pharmacokin. Biopharm. 10:53–75 (1982).
I. Odar-Cederloff and O. Borga. Kinetics of diphenylhydantoin in uremic patients: Consequences of decreased plasma protein binding.Eur. J. Clin. Pharmacol. 7:31–37 (1974).
D. Kurata and G. R. Wilkinson. Erythrocyte uptake and plasma binding of diphenylhydantoin.Clin. Pharmacol. Ther. 16:355–362 (1974).
D. D. Breimer, C. Honhoff, W. Zilly, E. Richter, and J. M. van Rossum. Pharmacokinetics of hexobarbital in man after intravenous infusion.J. Pharmacokin. Biopharm. 3:1–11 (1975).
W. Zilly, D. D. Breimer, and E. Richter. Hexobarbital disposition in compensated and recompensated cirrhosis of the liver.Clin. Pharmacol. Ther. 23:525–534 (1978).
Y. J. Lin, S. Awazu, M. Hanano, and H. Nogami. Pharmacokinetic aspects of elimination from plasma and distribution to brain and liver of barbiturates in rat.Chem. Pharm. Bull. (Tokyo) 21:2749–2756 (1973).
M. Ernebo. Pharmacokinetics and distribution properties of pentobarbital in humans following oral and intravenous administration.J. Pharm. Sci. 63:1114–1118 (1974).
M. Ehrnebo and I. Oder-Cederlof. Binding of amobarbital, pentobarbital and diphenylhydantoin to blood and plasma proteins in healthy volunteers and uraemic patients.Eur. J. Clin. Pharmacol. 8:445–453 (1975).
S. Toon and M. Rowland. Structure-pharmacokinetic relationships among the barbiturates in the rat.J. Pharmacol. Exp. Ther. 225:752–763 (1983).
H. B. Hucker. Effect of halofenate on binding of various drugs to human plasma protein and on the plasma half life of antipyrine in monkeys.J. Pharm. Sci. 61:1490–1492 (1972).
J. T. Slattery, G. Levy, A. Jain, and F. G. McMahon. Effect of naproxen on the kinetics of elimination and anticoagulant activity of a single dose of warfarin.Clin. Pharmacol. Ther. 25:51–66 (1979).
S. B. Matin, S. H. Wan, and J. H. Karam. Pharmacokinetics of tolbutamide: Prediction by concentration in saliva.Clin. Pharmacol. Ther. 16:1052–1057 (1974).
O. Sugita, Y. Sawada, Y. Sugiyama, T. Iga, and M. Hanano. Prediction of drug-drug interaction fromin vitro plasma protein binding and metabolism: A study of tolbutamidesulfonamide interaction in rats.Biochem. Pharmacol. 30:3347–3354 (1981).
U. Klotz, T. Rapp, and W. A. Muller. Disposition of valproic acid in patients with liver disease.Eur. J. Clin. Pharmacol. 13:55–60 (1978).
W. Loscher. Serum protein binding and pharmacokinetics of valproate in man, dog, rat and mouse.J. Pharmacol. Exp. Ther. 204:255–261 (1978).
A. J. Wilensky, P. N. Friel, R. H. Levy, C. P. Comfort and S. P. Kaluzny. Kinetics of phenobarbital in normal subjects and epileptic patients.Eur. J. Clin. Pharmacol. 23:87–92 (1982).
I. M. Patel, R. H. Levy, and R. E. Cutler. Phenobarbital-valproic acid interaction.Clin. Pharmacol. Ther. 27:32–36 (1980).
J. N. Mcarther, P. D. Dawkins, and M. J. H. Smith. The binding of indomethacin, salicylate and phenobarbital to human whole bloodin vitro.J. Pharm. Pharmacol. 23:32–36 (1971).
G. E. Mawer, N. E. Miller, and L. A. Turnberg. Metabolism of amylobarbitone in patients with chronic liver disease.Br. J. Pharmacol. 44:549–560 (1972).
K. Balasubramaniam, S. B. Lucas, G. E. Mawer, and P. J. Simons. The kinetics of amylobarbitone metabolism in healthy men and women.Br. J. Pharmacol. 39:564–572 (1970).
H. R. Oches, D. J. Greenblatt, E. Woo, K. Franke, and T. W. Smith. Effect of propranolol on pharmacokinetics and acute electrocardiographic changes following intravenous quinidine in humans.Pharmacology 17:301–306 (1978).
I. E. Hughes, K. E. Ilett, and I. B. Jellett. The distribution of quinidine in human blood.Br. J. Clin. Pharmacol. 3:521–525 (1975).
S. H. Curry. Relation between binding to plasma protein, apparent volume of distribution and rate constants of disposition and elimination for chlorpromazine in three species.J. Pharm. Pharmacol. 24:818–819 (1972).
M. H. Bickel. Binding of chlorpromazine and imipramine to red cells, albumin, lipoproteins and other blood components.J. Pharm. Pharmacol. 27:733–738 (1975).
M. Ehrnebo, S. Agurell, L. O. Boreus, E. Gordon, and U. Lonroth. Pentazocine binding to blood cells and plasma proteins.Clin. Pharmacol. Ther. 16:424–429 (1974).
E. A. Neal, P. J. Meffin, P. B. Gregory, and T. F. Blaschke. Enhanced bioavailabilty and decreased clearance of analgesics in patients with cirrhosis.Gastroenterology 77:96–102 (1979).
S. Agurell, L. O. Boreus, E. Gordon, J. E. Lindgrem, M. Ehrnebo, and U. Lonroth. Plasma and cerebrospinal fluid concentrations of pentazocine in patients: Assay by mass fragmentography.J. Pharm. Pharmacol. 26:1–8 (1974).
M. Ehrnebo, L. O. Boreus, and U. Lonroth. Bioavailability and first-pass metabolism of oral pentazocine in man.Clin. Pharmacol. Ther. 22:888–892 (1977).
Y. Igari, Y. Sugiyama, Y. Sawada, T. Iga, and M. Hanano.In vitro andin vivo assessment of hepatic and extrahepatic metabolism of diazepam in the rat.J. Pharm. Sci. 73:825–828 (1984).
T. Nakagawa, Y. Koyanagi, and H. Togawa. SALS, A Computer Program for Statistical Analysis with Least Squares Fitting. Library Program of University of Tokyo Computer Center, Tokyo, Japan (1978).
E. J. Triggs, R. L. Nation, A. Long, and J. J. Ashley. Pharmacokinetics in the elderly.Eur. J. Clin. Pharmacol. 8:55–62 (1975).
J. Aarbakke, O. M. Bakke, E. J. Milde, and D. S. Davies. Disposition and oxidation metabolism of phenylbutazone in man.Eur. J. Clin. Pharmacol. 11:359–366 (1977).
D. Horwitz, S. S. Thorgeirsson, and J. R. Mitchell. The influence of allopurinol and size of dose on the metabolism of phenylbutazone in patients with gout.Eur. J. Clin. Pharamcol. 12:133–136 (1977).
P. J. Neuvonen and E. E. Elonen. Effect of activated charcoal on absorption and elimination of phenobarbital, carbamazepine and phenylbutazone in man.Eur. J. Clin. Pharmacol. 17:51–57 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sawada, Y., Hanano, M., Sugiyama, Y. et al. Prediction of the disposition of nine weakly acidic and six weakly basic drugs in humans from pharmacokinetic parameters in rats. Journal of Pharmacokinetics and Biopharmaceutics 13, 477–492 (1985). https://doi.org/10.1007/BF01059331
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01059331