Abstract
Hydrolyzed olive pit residues from the production of furfural were ammoniated to obtain a nitrogenenriched humic fertilizer. Samples had been previously oxidized with nitric acid. The oxidized samples were further ammoniated in a batch reactor in the following range of experimental conditions: T = 80−210°C,\(C_{HNO_3 } = 0.5 - 5.0 wt\%\), solid/liquid ratio, S/L = 0.5−2.8 w/w and reaction time, t = 1−4 h.
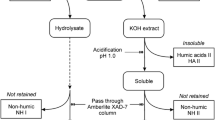
Planning of experiments was made by statistical design and results were fitted by multiple non-lineal regression polynomials. The product consists of two fractions: a water soluble fraction which contains most of the nitrogen (10−13 wt%) and a solid fraction with a very low nitrogen content (<4.0 wt%), which is likely to be fixed during the oxidation step. The soluble fraction can be used as a humic fertilizer and the solid product as a soil conditioner.
Similar content being viewed by others
References
Agatzini S and Burkin AR (1985) Statistical approach to the precipitation of iron as goethite. Trans Instn Min Metall 31: C105-C113
Baris H and Dincer C (1983) Lignite-based nitrogenous fertilizer. Energy Sources 7: 87–94
Berkowitz N, Chakrabartty SK, Cook FD and Fujikawa JI (1970) On the agrobiological activity of oxidatively ammoniated coal. Soil Sci 110: 211–217
Coca J, Alvarez R and Fuertes AB (1984) Production of nitrogenous humic fertilizer by oxidation-ammoniation of lignite. Ind Eng Chem Prod Res Dev 23: 620–624
Coca J, Alvarez R and Fuertes AB (1985) Oxiammoniation of pine-bark particles. Can J Chem Eng 63: 835–839
Davis ROE and Scholl W (1939) Ammoniated peat ⋯ Effect of varying the conditions of ammonia treatment on nitrogen quality. Ind Eng Chem 31: 185–189
Gnanasekaran A and Rao C (1978) Formation of furfural from rice hull in a fixed bed reactor. Indian Chem Eng 20: 3–8
Grethlein HE (1977) Statistical design of experiments for optimizing the casting variables for cellulose acetate membranes. In: Sourirajan S (ed) Reverse osmosis and synthetic membranes theory-technology-engineering. Canada
Gürüz K (1980) Oxiammoniation of Elbistan lignite to produce a nitrogenous fertilizer. Fuel 59: 772–77
Higuchi K and Asakawa K (1960) Ammonium humate manufactured from lignite. Japan Patent 8658 ('58)
Jaeggle W (1976) Integrated production of furfural and acetic acid from fibrous residues in a continuous process. Chem Age India 27: 521–530
Kim YK, Plain WM and Hatfield JD (1981) Fertilizer from the oxidative ammoniation of sawdust. Ind Eng Chem Prod Res Dev 20: 205–212
Kizer O, Laguerie C and Angelino H (1977) Etude experimentale de l'oxidation catalytique du bencene en anhydride maleique en couche fluidisee. Chem Eng J 14: 205–215
Kizer O, Chavarie C, Laguerie C and Cassimatis D (1978) Quadratic model of the behaviour of a fluidized bed reactor: Catalytic oxidation of benezene to maleic anhydride. Can J Chem Eng 56: 716–724
Mukherjee PN, Bhaumik JN and Lahiri A (1965) Production of fertilizers from coal. Indian J Technol 3: 90–92
Mukherjee PN, Banerjee S, Ramchandran LV and Lahiri A (1966) Production of fertilizers from coal. Study of reactions variables. Indian J Technol 4: 119–123
Nee CI and Yee WF (1976) Hydrolysis of pentosans in bagasse pith. J Appl Chem Biotechnol 26: 283–287
Official Methods of Analysis, AOAC (1975) William Horwitz (ed) Washington pp 15–19
Oka H, Inoue S and Sasaki M (1974) Studies on the reaction of peat humic acid with ammonia. Kitami Kogyo Tanki Daigaku Kenkyu 6: 132–138
Oka H, Inoue S and Sasaki M (1980) Reactions of peat humic acid with ammonia. Nenryo Kyokoishi 59: 241–249
Riera FA (1989) Aprovechamiento integral de biomasa residual: Obtención de furfural y fertilizantes húmicos nitrogenados. Ph.D. Dissertation. University of Oviedo
Scholl W and Davies ROE (1933) Ammoniation of peat for fertilizers. Ind Eng Chem 25: 1074–1078
Toynbee PA, Fleming AK (1963) Air oxidation of subbituminous coal. Fuel 42: 279–387
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Riera, F.A., Alvarez, R. & Coca, J. Humic fertilizers by oxiammoniation of hydrolyzed olive pits residues. Fertilizer Research 28, 341–348 (1991). https://doi.org/10.1007/BF01054335
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01054335