Abstract
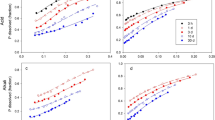
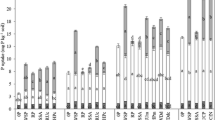
More than in the case of the other major fertiliser elements the performance of phosphorus in fertilisers in dependent on the state of chemical combination of the element. As distinct from typical American practice where fully acidulated fertilisers are emphasised, some other countries have moved in part to production of materials of less than full acidulation where some reliance is placed on the reactivity of the original phosphate rock. Solutions of various aliphatic acids and their salts have been used as tests for the availability of phosphorus in fully acidulated fertilisers and, to a degree, for phosphate rocks themselves. Less than fully acidulated products are at present assessed in the same way as fully acidulated materials, but in this and other evaluations problems and anomalies arise which are discussed within the framework of a number of fertiliser systems. Analytical data are adduced which illustrate the effects of some extraneous cations on the results achieved which conflict with some previous opinions. Research towards a fuller understanding of the subject is suggested.
Similar content being viewed by others
References
Barton AFM and McConnel SR (1975) Dissolution rate of a New Zealand Superphosphate
Barton AFM and McConnel SR (1979) Dissolution behaviour of solids: The rotating disc method. Chemin Aust 46: 427
Braithwaite AC (1987) The use of chemical solubility tests in comparing phosphate fertilisers. Fert Res 12: 185–192
Braithwaite AC, Eaton AC and Groom PS (1989) Some factors associated with the use of the extractants 2% citric acid and 2% formic acid as estimators of available phosphorus in fertiliser products. Fert Res 19: 181–195
Evans L (1978) Multiple extractions of phosphate materials. NZ FMRA Research Reports p 154–157
Ibid (1979) Multiple extractions of phosphate NZ FMRA Research Reports p 256–259
Higgins DJ (1982) Continuous methods on solubility of superphosphate. Proc N2 FMRA Res Symp 18–25
Hoffman WM and Brenn HJ (1964) Phosphate rock solubilization by repeated extractions with citrate solutions. J Ag & Food Chem 12: 344–346
Krum HE (1969) The limitations of citric solubility as a measure of availability of rock phosphate
Lehr JR and McClellan GH (1972) A revised laboratory reactivity scale for evaluating phosphate rocks for direct application. TVA Bulletin 7–43
Ibid (1967) Characterisation of apatites in commercial phosphate rocks. Colloque international sur les phosphates minereaux solides
McConnel SR (1981) Computor-acided determination of apatite unit cell parameters. NZ FMRA Research Reports p 175–181
Ibid (1983) Fertilizer dissolution kinetics—Part VII—‘Reactivity’ of phosphate rocks from various sources. NZ FMRA Research Reports p 177–185
McConnel SR and Evans L (1981) Dissolution rates of phosphate rocks using the stirred reactor. NZ FMRA Research Reports p 292–296; p 31–38; p 81–101; p 103–107; p 109–112
Ibid (1982). Fertilizer dissolution kinetics. Part VI. Flow through system. NZ FMRA Research Reports p 1–16
McKay AD, Syers JK and Gregg PEH (1984) Ability of chemical extraction procedures to assess the agronomic effectiveness of phosphate rock materials. NZJ Agric Res 27: 219–230
Quin BF (1982) Use of citric acid soluble and water soluble phosphate to assess the agronomic value of fertilisers. Proc NZ FMRA Res Symp p 40–54
Rogers DA (1984) The effect of stirring speed on the rate of dissolution of Naura phosphate rock. NZ FMRA Research Reports p 21–36; p 95–104
Steven RC (1982) The relationship between phosphorus solubility of single superphosphate in different extractants and the dry matter production of ryegrass in pots. Proc NZ FMRA Res Symp p 24–39
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Higgins, D.J., Braithwaite, A.C. The determination of phosphate availability for some less conventional phosphate fertilisers. Fertilizer Research 26, 271–280 (1990). https://doi.org/10.1007/BF01048765
Issue Date:
DOI: https://doi.org/10.1007/BF01048765