Abstract
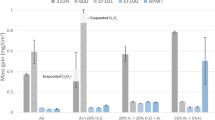
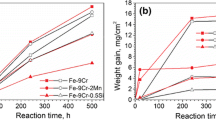
Oxidation tests of rare-earth-modified and Ti-modified Fe−20Cr alloy foils, which are under consideration for catalytic converter supports, were performed in a synthetic exhaust-gas atmosphere (N2+H2O+CO2) between 900°C and 650°C. Between 900°C and 750°C, the rare earths had no effect on oxide growth rates while Ti increased growth rates. Oxide growth rates for the rareearth alloys at 800°C and 750°C are much lower than those found in the literature for oxidation of Fe−Cr alloys or pure Cr in O2-rich atmospheres. The slow growth rates for the rare-earth alloys agree with literature data for oxidation of stainless steels containing >20% Cr in wet atmospheres and are caused by growth of an oxide scale only one grain thick. At temperatures ≤700°C, Fe−20Cr alloys grow massive Fe oxides; however, this can be suppressed by adding rare earths or Ti. To ensure good oxide adherence, free sulfur must be eliminated in the alloy by tying it up with a reactive-element addition. Both Ti and the rare earths can be used to tie up S, but the rare earths are more effective. For converter applications, the optimum alloy composition may contain rare earths for good oxide adherence and a small amount of Ti to suppress growth of Fe-rich oxides.
Similar content being viewed by others
References
D. R. Sigler,Oxid. Met. 29, 21–43 (1988).
A. W. Funkenbusch, J. G. Smeggil, and N. S. Bornstein,Mat. Trans. 16A, 1164–1166 (1985).
J. G. Smeggil, A. W. Funkenbusch, and N. S. Bornstein,Met. Trans. 17A, 923–932 (1986).
J. L. Smialek,Met. Trans. 18A, 164–167 (1987).
G. C. Wood and D. P. Whittle,Corros. Sci. 7, 763–782 (1967).
I. Kvernes, M. Oliveira, and P. Kofstad,Corros. Sci. 17, 237–252 (1977).
N. Birks and G. H. Meier,Introduction to High Temperature Oxidation of Metals (Edward Arnold, London, 1983), pp. 110–112.
N. Otsuka, Y. Shida, and H. Fujikawa,Oxid. Met. 32, 13–45 (1989).
Y. Ikeda and K. Nii,Trans. Nat. Res. Inst. Met. 26, 52–62 (1984).
C. T. Fujii and R. A. Meussner,J. Electrochem. Soc. 111, 1215–1221 (1964).
H. Hindam and D. P. Whittle,Oxid. Met. 18, 245–284 (1982).
E. J. Felten,J. Electrochem. Soc. 108, 490–495 (1961).
J. M. Francis and W. H. Whitlow,Corros. Sci. 5, 701–710 (1965).
E. Tsuzi,Met. Trans. 11A, 1965–1972 (1980).
G. C. Wood and J. Boustead,Corros. Sci. 8, 719–723 (1968).
F. I. Wei and F. H. Stott,Corros. Sci. 29, 839–861 (1989).
T. N. Rhys-Jones and H. J. Grabke,Mater. Sci. Technol. 4, 446–454 (1988).
D. R. Sigler,Oxid. Met. 32, 337–355 (1989).
I. Barin and O. Knacke,Thermochemical Properties of Inorganic Substances (Springer-Verlag, Berlin, 1973).
I. Barin, O. Knacke, and O. Kubaschewski,Thermochemical Properties of Inorganic Substances Supplement (Springer-Verlag, Berlin, 1977).
K. C. Mills,Thermodynamic Data for Inorganic Sulphides, Selenides, and Tellurides (Butterworths, London, 1974).
K. A. Gschneidner and N. Kippenhan,Thermochemistry of the Rare Earth Carbides, Nitrides, and Sulfides for Steelmaking (Rare Earth Information Center, Institute for Atomic Research, Iowa State University, Ames, Iowa, 1971).
J. F. Elliott, M. Gleiser, and V. Ramakrishna,Thermochemistry for Steelmaking, Vol. II (Addison-Wesley, Reading, MA, 1963), p. 520.
D. R. Sigler,Oxid. Met. 36, 57–80 (1991).
B.Pieraggi,Oxid. Met. 27, 177–185 (1987).
D. R. Sigler,Oxid. Met. 40, 555–583 (1993).
C. S. Tedmon,J. Electrochem. Soc. 114, 788–795 (1967).
G. C. Wood and D. P. Whittle,J. Electrochem. Soc. 115, 126–133 (1968).
D. Mortimer and W. B. A. Sharp,Br. Corros. J. 3, 61–67 (1968).
D. Caplan and G. I. Sproule,Oxid. Met. 9, 459–472 (1975).
E. A. Gulbransen and K. F. Andrew,J. Electrochem. Soc. 104, 334–338 (1957).
W. C. Hagel,Trans. ASM 56, 583–599 (1963).
C. S. Giggins and F. S. Pettit,Trans. Metall. Soc. AIME 245, 2495–2507 (1969).
C. A. Phalnikar, E. B. Evans, and W. M. Baldwin,J. Electrochem. Soc. 103, 429–438 (1956).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sigler, D.R. The oxidation behavior of Fe-20Cr alloy foils in a synthetic exhaust-gas atmosphere. Oxid Met 46, 335–364 (1996). https://doi.org/10.1007/BF01048635
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01048635