Abstract
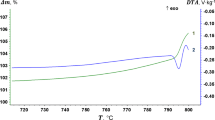
A set of independent methods has been used to study the catastrophic oxidation of copper in the system Cu−MexOy (where Me is Bi, W, Mo, or V). Two stages of the catastrophic oxidation have been revealed: a rapid stage (K∼10−4 kg2 m−4 sec−1) and a “super rapid” stage when the metal is oxidized within1–5 sec. The weight ratios of metal to oxidizer and the partial oxygen pressure for the superrapid copper oxidation have been established. The mechanism of the catastrophic oxidation of metals is considered.
Similar content being viewed by others
References
W. C. Leslie and M. G. Fontana,Trans. ASM. 43, 323 (1951).
V. V. Belousov,Oxid. Met. 38(3/4), 289 (1992).
V. V. Belousov,J. Superconduct. Phys. Chem. Tech. Russia 3(8), 1895 (1990).
C. Wagner,Z. Phys. Chem. B 32, 447 (1936).
J.-C. Boivin, O. Thomas, and G. Tridot,Comp. Rend. Acad. Sci. C 276, 1105 (1973).
H. Haas and E. Kordes,Z. Kristallogr. 129, 252 (1969).
M. M. Kohlmuller and J.-P. FaurieComp. Rend. Acad. Sci. C 264, 1751 (1976).
P. D. Robinson, J. M. Hughes, and M. L. Malinconico,J. Am. Mineral. 72, 397 (1987).
L. A. Onuchak,Structural Changes Investigation in Polycrystal Zn under Liquid Ga Contact, Ph.D., thesis (Moscow State University, Moscow, Russian, 1975).
L. A. Pogosyan,Moscow State Univ. Bull. (Chem.) 5 (1974).
A. L. Laskar and S. Chandra, eds., (Superionic Solids and Solid Electrolytes: Recent Trends) (Academic Press, New York, 1989).
Rights and permissions
About this article
Cite this article
Belousov, V.V. The kinetics and mechanism of catastrophic oxidation of metals. Oxid Met 42, 511–528 (1994). https://doi.org/10.1007/BF01046763
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01046763