Summary
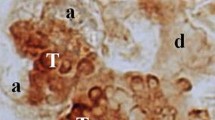
Nerve growth factor (NGF) was localized in the submandibular, sublingual, and parotid salivary glands of male and female diabetic mice and their normal littermates by immunoperoxidase staining usingp-phenylenediamine-pyrocatechol as a chromogen for the cytochemical demonstration of peroxidase activity. In the normal male submandibular gland, immunoreactive NGF was localized in the apical regions of granular, intercalated and collecting duct cells, while in the normal female submandibular gland, NGF was present throughout the cytoplasm of granular duct cells. The localization of NGF in the diabetic male and female submandibular glands was similar and resembled that of the normal female. NGF immunoreactivity was also observed in the striated duct cells in the sublingual and parotid glands of all four types of mice.
The sympathetic innervation of the submandibular glands of normal and diabetic mice was demonstrated using glyoxylic acid-induced histofluorescence. The pattern of sympathetic innervation and the intensity of catecholamine fluorescence was consistently different in the four types of mice. In the normal male submandibular gland the fluorescence was very intense, particularly in nerves adjacent to the granular ducts. In the normal female submandibular gland, the fluorescence was weak, while in the diabetic male and female the fluorescence was moderate.
The correlation between the intensity of the immunocytochemical staining for NGF and the catecholamine fluorescence adjacent to the granular ducts suggests a trophic influence of the NGF-containing granular ducts on their sympathetic innervation.
Similar content being viewed by others
References
Berkman, M. D. &Kronman, J. H. (1970) A histochemical study of the effects of castration and testosterone administration on the major salivary glands of Swiss Mice.Acta Anat., Berlin 76, 200–19.
Bocchini, V. &Angeletti, P. U. (1969) The nerve growth factor: Purification as a 30,000 Molecular weight protein.Proc. natn. Acad. Sci. U.S.A. 64, 787–94.
Bradshaw, R. A. (1978) Nerve growth factor.Ann. Rev. Biochem. 47, 191–216.
Carson, K. A., Hanker, J. S. & Kirshner, N. (1982) The adrenal medulla in mouse diabetes mellitus: Biochemical and morphological changes.Comp. Physiol. Biochem. (in press).
Chang, T. S., Erdos, E. G., Miwa, I., Tague, L. L. &Coalson, J. J. (1968) Isolation from a salivary gland of granules containing renin and kallikrein.Circ. Res. 23, 507–17.
Chrétien, M. (1977) Action of testosterone on the differentiation and secretory activity of a target organ: the submaxillary gland of the mouse.Int. Rev. Cytol. 50, 333–96.
Cohen, S. (1960) Purification of a nerve-growth promoting protein from the mouse salivary gland and its neurocytotoxic antiserum.Proc. natn. Acad. Sci., U.S.A. 46, 302–11.
Cohen, S. (1962) Isolation of a mouse submaxillary gland protein accelerating incisor eruption and eyelid opening in the new-born animal.J. biol. Chem. 237, 1555–62.
Coleman, D. &Hummell, K. (1967) Studies with the mutation diabetes in the mouse.Diabetologia 3, 238–48.
Coleman, D. &Hummel, K. (1968) The mutation diabetes in the mouse. InDiabetes. Proceedings of the Sixth Congress of the International Diabetes Federation (edited byOstman, J.), pp. 813–820. Amsterdam: Excerpta Medica.
De la Torre, J. C. &Surgeon, J. W. (1976) A methodological approach to rapid and sensitive monoamine histofluorescence using a modified glyoxylic acid technique: The SPG method.Histochemistry 49, 81–93.
Ekfors, T. &Hopsu-Havu, V. (1971) Immunofluorescent localization of trypsin-like estero-peptidases in the mouse submandibular gland.Histochem. J. 3, 415–20.
Fava-De-Moraes, F., Zangheri, E. O. &Doine, A. I. (1979) Immunohistochemical localization of erythropoietin in the rat and mouse submandibular gland.Histochem. J. 11, 97–102.
Giachetti, A. (1978) The functional state of sympathetic nerves in spontaneously diabetic mice.Diabetes 27, 969–74.
Goldstein, M. N. &Burdman, J. A. (1965) Studies of the nerve growth factor in the submandibular glands of female mice treated with testosterone.Anat. Rec. 151, 199–209.
Gresik, E. &Barka, T. (1977) Immunocytochemical localization of epidermal growth factor in mouse submandibular gland.J. Histochem. Cytochem. 25, 1027–35.
Gresik, E., Michelakis, A. &Barka, T. (1978) Immunocytochemical localization of renin in the submandibular gland of the mouse during postnatal development.Am. J. Anat. 153, 443–50.
Hanker, J. S., Yates, R. E., Metz, C. &Rustioni, A. (1977) A new, specific, sensitive, and noncarcinogenic reagent for the demonstration of horseradish peroxidase.Histochem. J. 9, 789–92.
Hanker, J. S., Carson, K. A., Yates, P. E., Preece, J. W., Doe, D. A., Ambrose, W. W. &Coffey, J. C. (1980a) Cytochemical correlates of structural sexual dimorphism in granular tissues of the mouse. II. Hypogonadic effects in the submandibular gland and kidney due to diabetes.Histochemistry 68, 99–118.
Hanker, J. S., Ambrose, W. W., Yates, P. E., Koch, G. G. &Carson, K. A. (1980b) Peripheral neuropathy in mouse hereditary diabetes mellitus. I. Comparison of neurologic, histologic, and morphometric parameters with dystonic mice.Acta Neuropath., Berlin 51, 145–54.
Harper, G. P., Al-Saffar, A. M., Pearce, F. L. &Vernon, C. A. (1980a) The production of nerve growth factorin vitro by tissues of the mouse, rat, and embryonic chick.Devl Biol. 77, 379–90.
Harper, G. A., Pearce, F. L. &Vernon, C. A. (1980b) The production and storage of nerve growth factorin vivo by tissues of the mouse, rat, guinea pig, hamster, and gerbil.Devl Biol. 77, 391–402.
Hummel, K., Dickie, M. &Coleman, D. (1966) Diabetes, a new mutation in the mouse.Science 153, 1127–28.
Junqueira, L. C. U., Fajer, A., Rabinovitch, M. &Frankenthal, L. (1949) Biochemical and histochemical observations on the sexual dimorphism of mice submaxillary glands.J. cell. comp. Physiol. 34, 129–58.
Kumar, S., Steward, J. K., Taylor, G. &Waghe, M. A. (1972) Fluorescence studies using anti-nerve growth factor.Expl Cell Res. 74, 170–4.
Levi-Montalcini, R. &Angeletti, P. U. (1961) Growth control of the sympathetic system by a specific protein factor.Q. Rev. Biol. 36, 99–108.
Levi-Montalcini, R. &Angeletti, P. U. (1968) Nerve growth factor.Physiol. Rev. 48, 534–69.
Levi-Montalcini, R. (1976) milestones, unanswered questions and current studies on nerve growth factor. InProceedings of the Sixth International Congress on Pharmacology (edited byAhtee, L.), pp. 221–230. Oxford: Pergamon Press.
Murphy, R. A., Saide, J. D., Blanchard, M. H. &Young, M. (1977) Nerve growth factor in mouse serum and saliva: Role of the submandibular gland.Proc. natn. Acad. Sci., U.S.A. 74, 2330–3.
Petrusz, P., Dimeo, P., Ordronneau, P., Weaver, C. &Keefer, D. (1975) Improved immunoglobulin enzyme bridge method for light microscopic demonstration of hormone-concentrating cells of the rat adenohypophysis.Histochemistry 46, 9–26.
Raynaud, J. (1960) Control hormonal de la glande sous-maxillaire de la souris.Bull. Biol. Fr. Belg. 94, 399–523.
Schwab, M. L. E., Stockel, K. &Thoenen, H. (1976) Immunocytochemical localization of nerve growth factor (NGF) in the submandibular gland of adult mice by light and electron microscopy.Cell Tissue Res. 169, 289–99.
Simson, J. A. V., Hazen, D., Spicer, S. S., Murphy, R. A. &Young, M., (1978) Secretogogue-mediated discharge of nerve growth factor from granular tubules of male mouse submandibular glands: An immunocytochemical study.Anat. Rec. 192, 375–88.
Sternberger, L. (1974)Immunocytochemistry, pp. 129–171. Englewood Cliffs, New York: Prentice-Hall.
Szpunar, W. E., Blair, A. J. &McCann, D. S. (1977) Plasma androgen concentrations in diabetic women.Diabetes 26, 1125–9.
Thoenen, H., Schwab, M. & Otten, U. (1978) Nerve growth factor as a mediator of information between effector organs and innervating neurons. InMolecular Control of Proliferation and Differentiation (edited byPapaconstantinou, J. andRitter, W.),Symposia of the Society for Developmental Biology Vol. 35, pp. 101–118.
Young, M., Murphy, R. A., Saide, J. D., Pantazis, N. J., Blanchard, M. H. &Arnason, B. G. W. (1976) Studies on the molecular properties of nerve growth factor and its cellular biosynthesis and secretion. InSurface Membrane Receptors (edited byBradshaw, R. A., Frazier, W. A., Merrell, R. C., Gottlieb, E. I. andHogue-Angeletti, R. H.), pp. 246–67. New York: Plenum Press.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Carson, K.A., Sar, M. & Hanker, J.S. Immunocytochemical demonstration of nerve growth factor and histofluorescence of catecholaminergic nerves in the salivary glands of diabetic mice. Histochem J 14, 35–48 (1982). https://doi.org/10.1007/BF01041129
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01041129