Summary
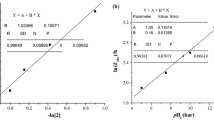
Di-μ-chlorobis(arylazooximato)dipalladiuni(II) compounds react with PPh3 to give the four-coordinate complexes (5) and (4) in which the azooxime acts as a unidentate and bidentate ligand respectively. Hydrogen bonding in polar solvents such as MeOH or EtOHetc. stabilize (5) whereas, polar solvents such as Me2CO, py and Et2O convert (5) into (4). The equilibrium: (4) + PPh3 ⇋ (5) exists in PhH solution and equilibrium constants at 30°C have been calculated spectrophotometrically. The variation in equilibrium constants and the stabilities of (5) have been explained on the basis of electron-releasing and electron-withdrawing properties of substituents, R, in the azooxime ligand.
Similar content being viewed by others
References
A. Chakravorty and K. C. Kalia,Inorg. Chem., 8, 2586 (1969).
P. K. Mascharak and A. Chakravorty,Ind. J. Chem., 18A, 471 (1979).
K. C. Kalia, A. Kumar, M. Singla and T. Kaur,Ind. J. Chem., 20A, 610 (1981).
E. Bamberger and W. Pemsel,Chem. Ber., 36, 85 (1903).
K. C. Kalia and A. Chakravorty,Inorg. Chem., 7, 2016 (1968);
B. Crociani, T. Boschi, R. Pietropaolo and U. Belluco,J. Chem. Soc. A, 531 (1970).
A. I. Vogel,A Textbook of Quantitative Inorganic Analysis, Longmans, London, 3rd Edit. 1961, p. 512.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kalia, K.C., Singla, M. & Kumar, A. The reaction of PPh3 with Di-μ-chlorobis(arylazooximato)dipalladium(II). Transition Met Chem 7, 151–153 (1982). https://doi.org/10.1007/BF01035830
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01035830