Abstract
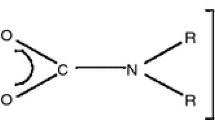
Both dimethylamino- (I) and diethylamino-alane dimers (II), [(R2N)2AlH]2 (I, R=Me; II, R=Et) decompose above 800° C under a few hundred Pa of hydrogen to dark greyish, hard (Vickers hardness larger than 2000), oxidation-resistive and oxidation-protective deposits which are tentatively identified as Al5C3N. The deposits are stable to moisture and diluted hydrochloric acid in contrast to those obtained below 800° C, but they easily dissolve in sodium hydroxide solution at room temperature evolving a gas. The deposits on a stainless steel substrate adhere strongly to the substrate and remain so on rapid heating and cooling. The electrical resistivity of the deposits is in the range 102 to 104 Ω cm.
Similar content being viewed by others
References
Y. Takahashi, K. Yamashita, S. Motojima andK. Sugiyama,Surf. Sci. 86 (1979) 238.
J. K. Ruff,J. Amer. Chem. Soc. 83 (1961) 2835.
R. A. Kovar andE. C. Ashby,Inorg. Chem. 10 (1971) 893.
“Diagrams and Tables for Quantitative Electron Probe Microanalysis” (Toyota Central R & D Labs., Inc. Nagoya, 1970).
G. A. Jeffrey andV. Y. Wu,Acta Cryst. 16 (1963) 559.
Idem, ibid. 20 (1966) 538.
C. D. Wagner,Anal. Chem. 44 (1972) 1050.
J. Czochralski,Z. Metallkunde 14 (1922) 278.
D. W. Lewis,J. Electrochem. Soc. 117 (1970) 978.
K. M. Taylor andC. Lenie,ibid. 107 (1960) 308.
R. W. Rice in “The Science of Hardness Testing and Its Research Applications”, edited by J. H. Westbrook and H. Conrad (American Society for Metals, Metals Park, Ohio, 1973) p. 117.
E. Ryshkewitch, “Oxide Ceramics: Physical Chemistry and Technology” (Academic Press, New York, 1960).
H. M. Manasevit, F. M. Erdmann andW. I. Simpson,J. Electrochem. Soc. 118 (1971) 1864.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Takahashi, Y., Mutoh, K., Motojima, S. et al. Further investigation on the thermal decomposition of aluminium dialkylamides. J Mater Sci 16, 1217–1222 (1981). https://doi.org/10.1007/BF01033835
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01033835