Summary
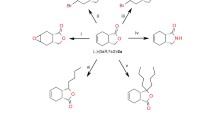
A method for the quantitative enantioselective bioreduction of ethyl acetoacetate [1] to optically pure (+)-S-ethyl-3 hydroxybutyrate [II] usingSclerotium rolfsii mycelium is described. In a synthetic medium 1 g mycelium (dry weight) could convert 1 g of I to II within 2–3 days of fermentation (pH 5.8, 30°C). This is the first report demonstrating use ofS. rolfsii biomass for asymmetric reduction to get chiral building blocks.
Similar content being viewed by others
References
Bucciarelli, M., Forni, A., Morretti, I. and Torre, H. (1983) Synthesis, No. 10, 897–899.
Fráter, G. (1979) Helv. Chim. Acta62, 2825–2828.
Georg, T., Gill, H.S. and Gerhardt, C. (1985) Tetrahedron Lett.26, 3903–3906.
Kelkar, H.S., Lachke, A.H. and Deshpande, M.V. (1988) Can. J. Microbiol.34, 83–85.
Lachke, A.H. and Deshpande, M.V. (1988) FEMS Microbiol. Reviews54, 177–193.
Mandels, M. and Weber, J. (1969) In: Cellulases and their applications (Gould, R.F. ed.) Adv. Chem. Ser.95, p. 391–414, Am. Chem. Soc., Washington, D.C.
Meyers, A.I. and Amos, R.A. (1980) J. Am. Chem. Soc.102, 870–872.
Mori, K. and Puapoomchareon, P. (1987) Liebigs Ann. Chem. 271–272
Mori, K. and Sakai, T. (1988) Liebigs Ann. Chem. 13–17.
Wipf, B., Kupfer, E., Bertazzi, R. and Wilhem Leuenberger, H.G. (1983) Helv. Chim. Acta66, 485–488.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wakharkar, R.D., Chanda, B. & Lachke, A.H. Microbial reduction of ethyl acetoacetate using Sclerotium rolfsii. Biotechnol Lett 11, 483–486 (1989). https://doi.org/10.1007/BF01026646
Issue Date:
DOI: https://doi.org/10.1007/BF01026646