Summary
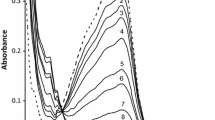
The interaction of aquo-ethylenediaminetetraacetatoruthenate(III) with ferricyanide ion was studied spectrophotometrically as a function of ferricyanide ion concentration, pH (1.5–8.5) and temperature (30–45°C) at ionic strength 0.2 M (NaClO4). Kinetic and activation parameters (ΔH≠=27.1±1.75 KJ mol−1, ΔS≠=−136.7±5.57 J mol−1 deg−1) are consistent with the proposed mechanism.
Similar content being viewed by others
References
K. Shimizu,Bull. Chem. Soc. Jpn.,50, 2921 (1977).
T. Matsubara and C. Cruetz,Inorg. Chem.,18, 1956 (1979).
H. C. Bajaj and R. V. Eldik,Inorg. Chem.,22, 4052 (1988).
H. C. Bajaj and R. V. Eldik,Inorg. Chem.,28, 1980 (1989).
A. A. Diamantis and J. V. Dubrawsky,Inorg. Chem.,20, 1142 (1981).
M. M. Taqui Khan, Amjad Hussain, G. Ramachandraiah and M. A. Moiz,Inorg. Chem.,25, 3023 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chatterjee, D., Ghosh, A.K. Kinetics of the interaction of aquo-ethylenediaminetetraacetatoruthenate(III) with ferricyanide ion in water. Transition Met Chem 16, 481–483 (1991). https://doi.org/10.1007/BF01024311
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01024311