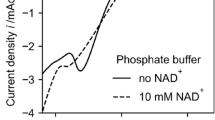
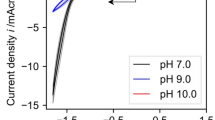
Summary
Enzymatically active NADH was formed from NAD+ directly in the electrochemical system by adopting an anion-charged membrane as a diaphragm and applying the less cathodic potential of −0.7V vs. Ag/AgCl electrode. The direct electrochemical procedure worked favorably as an NADH regenerator in the biochemical reaction system using D-lactate dehydrogenase which transforms pyruvate to D-lactate.
Similar content being viewed by others
References
Aizawa, M., Suzuki, S. and Kubo, M. (1976).Biochim. Biophys. Acta 444, 886–892.
Burnett, J. N. and Underwood, A. L. (1965).J. Org. Chem. 30, 1154–1158.
Cunningham, A. J. and Underwood, A. L. (1967).Biochem. 6, 266–271.
Dicosimo, R., Wong, C. H., Daniels, L. and Whitesides, G. M. (1981).J. Org. Chem. 46, 4623–4625.
Jaegfeldt, H. (1981).Bioelectrochem. Bioenerg. 8, 355–361.
Maeda, H. and Kajiwara, S. (1985).Biotechnol. Bioeng. 27, 596–602.
Nakamura A., Minami, H., Urabe, I. and Okada, H. (1988).J. Ferment. Technol. 66, 267–272.
Shaked, Z., Barber, J. J. and Whitesides, G. M. (1981).J. Org. Chem. 46, 4101–4103.
Sugimura, K., Kuwabata, S. and Yoneyama, H. (1990).Bioelectrochem. Bioenerg. 24, 241–247.
Vandecasteele, J. (1981).Appl. Environ. Microbiol. 39, 327–334.
Wichmann, R., Wandrey, C., Kula, M. R. and Buckmann, A. (1981).Biotechnol. Bioeng. 23, 2789–2802.
Wong, C. H. and Whitesides, G. M. (1981).J. Am. Chem. Soc. 103, 4890–4899.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yun, SE., Taya, M. & Tone, S. Direct reduction of NAD+ by electrochemical procedure and application of the regenerated NADH to enzyme reaction. Biotechnol Lett 16, 1053–1058 (1994). https://doi.org/10.1007/BF01022402
Issue Date:
DOI: https://doi.org/10.1007/BF01022402