Abstract
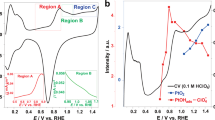
The electrooxidation of methanol was enhanced on PtSn-SPE, PtRu-SPE and PtIr-SPE in sulfuric acid solution, when compared with the activity of Pt-SPE, which has already been shown to have a higher activity than a Pt electrode. SPE is an abbreviation for Nafion, a solid polymer electrolyte. It is suggested that this dual enhancement of the oxidation rate for PtSn-SPE and PtRu-SPE catalysts is due to the modification of the oxidation state of Pt by Sn and Ru and to the presence of H2O and CH3OH, both modified by the SPE matrix. This modification appears to weaken their hydrogen bonds in solution. Both Pt and Ir have catalytic properties for methanol oxidation, but a PtIr-SPE catalyst showed a more enhanced catalytic activity than either of them. This will be discussed in terms of Ir, oxidized at relatively low positive potentials, assisting the redox process of Pt0/Pt2+ or Pt2+/Pt4+ in the SPE matrix, where CH3OH and H2O are present in modified forms. For comparison, IrPd-SPE was also used as an electrode and showed a higher activity than Ir alone, although Pd did not have any activity toward methanol oxidation in sulfuric acid solution. Irrespective of the kind of Pt-SPEs, the Tafel slope was approximately 120 mV; the CH3OH concentration dependence was of the order of 0.2–0.6. The pH dependence was nearly 0.5 against NHE. The activation energy of the Pt-SPEs for the reaction ranged between 20 and 33 kJ mol−1.
Similar content being viewed by others
References
P. W. Lu and S. Srinivasan,J. Appl. Electrochem. 9 (1979) 269.
R. S. Yeo,ACS Symp. Ser. 180 (1982) 453.
B. K. Kipling,ACS Symp. Ser. 180 (1982) 479.
I. Rubinstein and A. J. Bard,J. Am. Chem. Soc. 103 (1981) 5007.
N. Oyama, T. Simomura, K. Shigehara and F. C. Anson,J. Electroanal. Chem. 112 (1980) 271.
H. S. White, J. Leddy and A. J. Bard,J. Am. Chem. Soc. 104 (1982) 4811.
H. Nakajima, Y. Takabuwa, H. Kikuchi, K. Fujikawa and M. Kita,Electrochim. Acta. 32 (1987) 791.
K. A. Mauritz, C. J. Hora and A. J. Hopfinger,Advs. Chem. Ser. ACS 187 (1980) 123.
T. D. Gierke and W. Y. Hsu,ACS Symp. Ser. 180 (1982) 283.
In Ref. [9] the void cages are called clusters.
M. Falk,Can. J. Chem. 58 (1980) 1495.
M. Falk,ACS Symp. Ser. 180 (1982) 139.
W. G. F. Grot, G. E. Munn and P. N. Walmsley,Extended Abstracts, 141st Meeting of Electrochem. Soc., Houston (1972).
H. Takenaka and E. Torikai,Kokai Tokkyo Koho (Japan Patent)55 (1980) 38934.
V. S. Bagotzky and Yu. B. Vassiliev,Electrochim. Acta 12 (1967) 1323.
B. B. Damaskin, O. A. Petrii and V. V. Batrakov, ‘Adsorption of Organic Compounds on Electrodes’, Penum Press, New York (1971) Chaps 8 and 10.
M. W. Breiter,Electrochim. Acta 8 (1963) 973.
J. O'M. Bockris and H. Wroblowa,J. Electroanal. Chem. 7 (1964) 428.
K. J. Cathro,J. Electrochem. Soc. 116 (1969) 1608.
M. M. P. Janssen and J. Moolhuysen,Electrochim. Acta 21 (1976) 861, 869.
M. M. P. Janssen and J. Moolhuysen,J. Catalysis 46 (1977) 289.
A. Katayama,J. Phys. Chem. 84 (1980) 376.
A. Aramata and R. Ohnishi,J. Am. Chem. Soc. 105 (1983) 658.
A. Aramata and R. Ohnishi,J. Electroanal. Chem. 162 (1984) 153.
R. Woods,Israel J. Chem. 18 (1979) 118.
A. Aramata, T. Yamazaki, K. Kunimatsu and M. Enyo,J. Phys. Chem. 91 (1985) 2309.
L. Hilaire, G. D. Guerrero, P. Legare, G. Maire and G. Krill,Surf. Sci. 146 (1984) 569.
A. Aramata and I. Toyoshima,J. Electroanal. Chem. 135 (1982) 111.
H. Kita and A. Aramata, ‘Electrochemical Reactors’ (edited by M. Ismail), Elsevier, Amsterdam, Vol. 1, Chap. 3, in press.
K. Ota, Y. Nakagawa and M. Takahashi,J. Electroanal. Chem. 179 (1984) 179.
S. Glasstone, K. J. Laidler and H. Eyring, ‘The Theory of Rate Processes’, McGraw-Hill, NY (1941) p. 376.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aramata, A., Kodera, T. & Masuda, M. Electrooxidation of methanol on platinum bonded to the solid polymer electrolyte, Nafion. J Appl Electrochem 18, 577–582 (1988). https://doi.org/10.1007/BF01022253
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01022253