Abstract
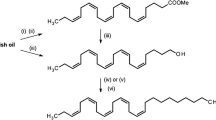
Unsaturated aliphatic pheromones ofH. virescens were prepared at high specific activity (3H, 58 Ci/mmol) and were employed to study tissue specificity of acetate esterase, alcohol oxidase, and aldehyde dehydrogenase in male and femaleHeliothis virescens. Thus, [9,10-3H2]Z9-14:Ac was synthesized by partial tritiation of the corresponding alkyne and was converted to the labeledZ9-14∶OH andZ9-14∶Al for metabolic studies. Soluble and membrane-associated enzyme activities were determined by radio-TLC assays. Esterase activity is highest in legs of both sexes, but also occurs in antennal and glandular tissues. Oxidase activity requires O2 and is highest in female pheromone gland tissues, but it is also high in the male hairpencils. Aldehyde dehydrogenase activity was uniformly high in all tissues, but highest in antennal tissues of both males and females.
Similar content being viewed by others
References
Bjostad, L.B., andRoelofs, W.L. 1981. Sex pheromone biosynthesis from radiolabeled fatty acids in the redbanded leafroller moth.J. Biol. Chem. 256:7935–7940.
Bjostad, L.B., andRoelofs, W.L. 1986. Sex pheromone biosynthesis in the red-banded leafroller moth studied by mass-labeling with stable isotopes and chemical ionization mass spectrometry.J. Chem. Ecol., this volume.
Bjostad, L.B., Wolf, W.A., andRoelofs, W.L. 1981. Total lipid analysis of the sex pheromone gland of the redbanded leafroller mothArgyrotinia velutinana, with references to pheromone biosynthesis.Insect Biochem. 11:73–79.
Blomquist, G.J., andDillwith, J.W. 1983. Pheromones: Biochemistry and physiology, pp 527–542,in R.G.H. Downer and H. Laufer, (eds.).Endocrinology of Insects. Alan R. Liss, New York.
Bradford, M.M. 1976. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding.Anal. Biochem. 72:248–254.
Charlton, R.E., andCardé, R.T. 1982. Rate and diel periodicity of pheromone emission from female gypsy moths (Lymantria dispar) determined with a glass-adsorption collection system.J. Insect Physiol. 28:423–430.
Ferkovich, S.M. 1981. Enzymatic alteration of insect pheromones, pp. 167–188,in D.M. Norris (ed.). Perception of Behavioral Chemicals. Elsevier/North Holland, Amsterdam.
Ferkovich, S.M., Mayer, M.S., andRutter, R.R. 1973. Conversion of the sex pheromone of the cabbage looper.Nature 242:53–55.
Ferkovich, S.M., Oliver, J.E., andDillard, C. 1982a. Comparison of pheromone hydrolysis by the antennae with other tissues after adult eclosion in the cabbage looper moth,Trichoplusia ni.Entomol. Exp. Appl. 31:327–328.
Ferkovich, S.M., Oliver, J.E., andDillard, C. 1982b. Pheromone hydrolysis by cuticular and interior esterases of the antennae, legs, and wings of the cabbage looper moth,Trichoplusia ni (Hubner).J. Chem. Ecol. 8:859–866.
Hammock, B.D., Wing, K.D., McLaughlin, J., Lovell, V., andSparks, T.C. 1982. Trifluoromethyl ketones as possible transition state analog inhibitors of juvenile hormone esterase.Pest. Biochem. Physiol. 17:76–88.
Hansen, K. 1984. Discrimination and production of disparlure enantiomers by the gypsy moth and the nun moth.Physiol. Entomol. 9:9–18.
Henrick, C.A. 1977. The synthesis of insect sex pheromones.Tetrahedron 34:1845–1889.
Kasang, G. 1974. Uptake of the sex pheromone [3H]bombykol and related compounds by male and femaleBombyx antennae.J. Insect Physiol. 20:2407–2422.
Kasang, G., Schneider, D., andBeroza, M. 1974a. Biosynthesis of the sex pheromone disparlure by olefin-epoxide conversion.Naturwissenschaften 61:13–131.
Kasang, G., Knauer, B., andBeroza, M. 1974b. Uptake of the sex attractant3H-disparlure by male gypsy moth antennae (Lymantria dispar=Porthetria dispar).Experientia 30:147–148.
Lonergan, G. 1986. Metabolism of pheromone components and analogs by cuticular enzymes ofChoristoneura fumiferana. J. Chem. Ecol. this volume.
Mayer, M.S., Ferkovich, S.M., andRutter, R.R. 1976. Localization and reactions of a pheromone degradative enzyme isolated from an insect antenna.Chemical Senses and Flavor 2:52–61.
Mayer, M.S., andMankin, R.W. 1985. Neurobiology of pheromone perceptionin J. Kerkut and L.T. Gilbert (eds.). Comprehensive Insect Physiology, Biochemistry, and Pharmacology, pp. 96–144, vol. 9, Pergamon Press, Oxford.
Meighen, E.A., Slessor, K.N., andGrant, G.G. 1981. A bioluminescence assay for aldehyde sex pheromones of insects.Experientia 37:555–556.
Morse, D., andMeighen, E. 1984a. Detection of pheromone biosynthetic and degradative enzymes in vitro.J. Biol. Chem. 259:475–480.
Morse, D., andMeighen, E. 1984b. Aldehyde pheromones in Lepidoptera: Evidence for an acetate ester precursor inChoristoneura fumiferana.Science 226:1434–1436.
Morse, D., andMeighen, E. 1986. Pheromone biosynthesis and the role of functional groups in pheromone specificity.J. Chem. Ecol. this volume.
Prestwich, G.D., Golec, F.A., andAndersen, N.H. 1984. Synthesis of a highly tritiated photoaffinity labelled pheromone analog for the mothAntheraea polyphemus.J. Lab. Compd. Radiopharmacol. 7:593–599.
Prestwich, G.D., Yamaoka, R., andCarvalho, J.F. 1985. Metabolism of tritiated ω-fluorofatty acids and alcohols in the termiteReticulitermes flavipes Kollar (Isoptera: Rhinotermitidae).Insect Biochem. 15:205–209.
Prestwich, G.D.,Vogt, R.G., andRiddiford, L.M. 1986. Binding and hydrolysis of radiolabeled pheromone and several analogs by male-specific antennal proteins of the mothAntheraea polyphemus. J. Chem. Ecol., this volume.
Raina, A.K., andKlun, J.A. 1984. Brain factor control of sex pheromone production in the female corn earworm moth.Science 225:531–533.
Richerson, J.V., andCameron, E.A. 1974. Differences in pheromone release and sexual behavior between laboratory-reared and wild gypsy moth adults.Environ. Entomol. 3:475–481.
Roelofs, W., andBjostad, L. 1984. Biosynthesis of lepidopteran pheromones.Bioorg. Chem. 12:279–298.
Sheads, R.E., andBeroza, M. 1973. Preparation of tritium-labeled disparlure, the sex attractant of the gypsy moth.J. Agric. Food Chem. 21:751–753.
Teal, P.E.A., andTumlinson, J.H. 1986. Induced in vivo biosynthesis of bombykal by sex pheromone glands ofHeliothis virescens andH. zea. J. Chem. Ecol., this volume.
Vogt, R.G. 1984. The biochemical design of sex pheromone reception in the wild silk mothAntheraea polyphemus. Dissertation. (University of Washington, Seattle).
Vogt, R.G., andRiddiford, L.M. 1980. Pheromone deactivation by antennal proteins of Lepidoptera, pp. 955–967,in F. Sehnal, A. Zabza, J. Menn, B. Cymborowski, (eds.). Regulation of Insect Development and Behavior. Wroclaw Technical University Press, Karpacz, Poland.
Vogt, R.G., andRiddiford, L.M. 1981. Pheromone binding and inactivation by moth antennae.Nature 293:161–163.
Vogt, R.G., andRiddiford, L.M. 1986. Pheromone reception: A kinetic equilibrium pp. 201–208,in T. Payne, M. Birch, and J. Kennedy (eds.). Mechanisms of Perception and Orientation to Insect Olfactory Signals. Oxford, England.
Vogt, R.G., andRiddiford, L.M. 1986. Scale esterase: A pheromone-degrading enzyme from the scales of the silk mothAntheraea polyphemus. J. Chem. Ecol., this volume.
Vogt, R.G., Riddiford, L.M., andPrestwich, G.D. 1985. Kinetic properties of a pheromonedegrading enzyme: The sensillar esterase ofAntheraea polyphemus, Proc. Natl. Acad. Sci. 82:8827–8831.
Yamaoka, R., Taniguchi, Y., andHayashiya, K. 1984. Bombykol biosynthesis from deuteriumlabeled (Z)-11-hexadecenoic acid.Experientia 40:80–81.
Author information
Authors and Affiliations
Additional information
Fellow of the Alfred P. Sloan Foundation (1981–1985) and Camille and Henry Dreyfus Teacher-Scholar (1981–1986).
Rights and permissions
About this article
Cite this article
Ding, Y.S., Prestwich, G.D. Metabolic transformation of tritium-labeled pheromone by tissues ofHeliothis virescens moths. J Chem Ecol 12, 411–429 (1986). https://doi.org/10.1007/BF01020564
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01020564