Abstract
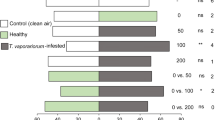
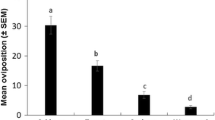
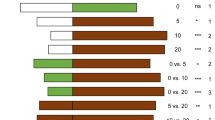
Cage experiments revealed that accessions of the wild tomato speciesLycopersicon hirsutum were preferred sites for oviposition byHeliothis zea. Hexane extracts from the leaves ofL. hirsutum were also preferred sites of oviposition in choice experiments among extracts from severalLycopersicon species. Extracts ofL. hirsutum were still biologically active several days after application, indicating that the phytochemical(s) involved are relatively stable and of low volatility. Gas Chromatographic analysis of leaf hexane extracts from 12 different accessions of theL. hirsutum complex and three tomato cultivars revealed substantial qualitative and quantitative variation in the chemical composition of these extracts. Comparison of these results with extract oviposition studies implicate a group of structurally related compounds as the active agents. Mass spectroscopy has tentatively identified these compounds as sesquiterpenes with the chemical formula C15H22O2. These compounds are apparently synthesized and secreted from glandular trichomes on the leaf surface. These phytochemicals did not stimulate ovipositional behavior in females of the cabbage looper,Trichoplusia ni. The existence of genetic variation for the presence and amount of kairomones that serve as cues for insect orientation and oviposition could be utilized in a breeding program to develop tomato cultivars with genetically modified allelochemic profiles that would disrupt the sequential behavioral processes of insect host-plant selection.
Similar content being viewed by others
References
Alvarado-Rodriguez, B., Leigh, T.F., andLange, W.H. 1982. Oviposition site preference by the tomato fruitworm (Lepidoptera: Noctuidae) on tomato, with notes on plant phenology.J. Econ. Entomol. 75:895–898.
Bordner, J., Danehower, D.A., Thacker, J.D., Kennedy, G.G., Stinner, R.E., andWilson, K.G. 1983. Chemical basis for host plant selection, pp. 245–264,in P.A. Hedin (ed.). Plant Resistance to Insects. ACS Symposium Series 208, American Chemical Society, Washington, D.C.
Burton, R.L., andSchuster, D.J. 1981. Oviposition stimulant for tomato pinworms from surfaces of tomato plants.Ann. Entomol. Soc. Am. 74:512–515.
Buttery, R.G., Ling, L.C., andChan, B.G. 1978. Volatiles of corn kernels and husks: Possible corn earworm attractants.J. Agric. Food Chem. 28:866–869.
Buttery, R.G., Ling, L.C., andTeranishi, R. 1980. Volatiles of corn tassels: Possible corn earworm attractants.J. Agric. Food Chem. 28:771–774.
Centello, W.W., andJacobsen, M. 1979. Corn silk volatiles attract many species of moths.J. Environ. Sci. Health. A14:695–709.
Coates, R.M.,Denissen, J.F.,Juvik, J.A., andBabka, B.A. 1988. Identification of α-santalenoic andendo-bergamotenoic acids as insect oviposition stimulants from wild tomato leaves.J. Org. Chem. 53 (in press).
Dimock, M.B., Kennedy, G.G. 1983. The role of glandular trichomes in the resistance ofLycopersicon hirsutum f.glabratum toHeliothis zea.Entomol. Exp. Appl. 33:263–268.
Duffey, S.S., andIsman, M.B. 1981. Inhibition of insect larval growth by phenolics in glandular trichomes of tomato leaves.Experientia 37:574–576.
Flath, R.A., Forreg, R.R., John, J.O., andChan, B.G. 1978. Volatile components of corn silk (Zeamays L.): PossibleHeliothis tea (Boddie) attractants.J. Agric. Food Chem. 26:1290–1293.
Hedin, P.A., Maxwell, F.G., andJenkins, J.N. 1974. Insect plant attractants, feeding stimulants, repellents, deterrents, and other related factors affecting insect behavior, pp. 527–594,in F.G. Maxwell and F.A. Harris (eds.). Proceedings, Summer Institute for Biological Control of Plant Insects and Disease. University Press of Mississippi, Jackson.
Johnson, M.W., Stinner, R.E., andRabb, R.L. 1975. Oviposition response ofHeliothis zea (Boddie) to its major hosts in North Carolina.Environ. Entomol. 4:291–297.
Jones, R.L., andBurton, R.L., McGovern, T.P., andBeroza, M. 1973. Potential oviposition inducers for corn earworms.Environ. Entomol. 66:921–925.
Juvik, J.A., Berlinger, M.J., Ben-David, T., andRudich, J. 1982. Resistance among accessions of the genusLycopersicon andSolanum to four of the major insect pests of tomatoes in Israel.Phytoparasitica 10:145–156.
Kennedy, J.S. 1977. Olfactory responses of insects to distant plants and other odor sources, pp. 69–91, in H.H. Shorey and J.J. McKelvey (eds.). Chemical Control of Insect Behavior. John Wiley & Sons, New York.
Luckwell, L.C. 1943. The genusLycopersicon: An historical, biological, and taxonomic survey of the wild and cultivated tomatoes. Aberdeen University Studies No. 120. Aberdeen University Press, Aberdeen, Scotland. 44 pp.
Lukefahr, M.J., Houghtailing, J.E., andGraham, H.M. 1971. Suppression ofHeliothis populations with glabrous cotton strains.J. Econ. Entomol. 64:486–488.
Lundgren, L., Norelius, G., andStenhagen, G. 1985. Leaf volatiles from some wild tomato species.Nord. J. Bot. 5:315–320.
Matsumoto, T. 1970. Volatile organic sulfur compounds as insect attractants with special reference to host selection, pp. 133–158,in D. Wood, R. Silverstein, and N. Nakajima (eds.). Control of Insect Behavior by Natural Products. Academic Press, New York.
Meisner, J., Ascher, K.R.S., andLavie, D. 1974. Factors influencing the attraction to oviposition of the potato tuber moth,Onorimoschema operculella Zell.Z. Angew. Entomol. 77:179–189.
Metcalf, R.L., Rhodes, A.M., Metcalf, R.A., Ferguson, J., Metcalf, E.R., andLa, P.-Y. 1982. Cucurbitacin contents and Diabroticite (Coleoptera: Chrysomelidae) feeding uponCucurbita spp.Environ. Entomol. 11:931–937.
Mitchell, N.D. 1977. Differential host selection byPieris brassicae L. (the large white butterfly) onBrassicae oleracea subsp. oleracea (the wild cabbage).Entomol. Exp. Appl. 21:283–389.
Moshonas, M.G., andLund, E.D. 1970. The mass spectra of sesquiterpene hydrocarbons.Flavor Ind. June:375–378.
Patterson, C.G., Knavel, D.E., Kemp, T.R., andRodriguez, J.G. 1975. Chemical basis for resistance byTetranychus urticae Koch in tomatoes.Environ. Entomol. 4:670–674.
Rick, C.M. 1973. Potential genetic resources in tomato species; clues from observations in native habitats, pp. 255–269,in A. Hollaender et al. (eds.). Genes, Enzymes, and Populations. Plenum Press, New York.
Schoonhoven, L.M. 1972. Plant recognition by lepidopterous larvae.Symp. R. Entomol. Soc. London 6:87–99.
Snodderly, L.J., andLambdin, P.L. 1982. Oviposition and feeding sites ofHeliothis zea on tomato.Environ. Entomol. 11:513–515.
Snyder, J.C., andCarter, C.D. 1984. Leaf trichomes and resistance ofLycopersicon hirsutum andL. esculentum to spider mites (Tetranychus urticae).J. Am. Soc. Hortic. Sci. 109:837–843.
Tichenor, L.H., andSeigler, D.S. 1980. Electroantennogram and oviposition responses ofManduca sexta to volatile components of tobacco and tomato.J. Insect Physiol. 56:309–314.
Visser, J.H., andAvé, D.A. 1978. General green leaf volatiles in the olfactory orientation of the Colorado potato beetle,Leptinotarsa decemlineata.Entomol. Exp. Appl. 24:534–549.
Walbauer, G.P., Cohen, R.W., andFriedman, S. 1984. An improved procedure for laboratory rearing of the corn earworm.Heliothis zea (Lepidoptera: Noctuidae).Great Lakes Entomol. 17:113–118.
Widstrom, N.W., McMillian, W.W., andWiseman, B.R. 1979. Ovipositional preference of the corn earworm and the development of trichomes on two exotic corn selections.Environ. Entomol. 8:833–839.
Williams, W.G., Kennedy, G.G., Yamamoto, R.T., Thacker, J.D., andBordner, J. 1980. 2- Tridecanone: A naturally occurring insecticide from the wild tomatoLycopersicon hirsutum f.glabratum. Science 207:888–889.
Yamamoto, R.T., andFraenkel, G. 1959. Common attractant for the tobacco hornworm and the Colorado potato beetle.Nature 184:206–207.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Juvik, J.A., Babka, B.A. & Timmermann, E.A. Influence of trichome exudates from species ofLycopersicon on oviposition behavior ofHeliothis zea (Boddie). J Chem Ecol 14, 1261–1278 (1988). https://doi.org/10.1007/BF01019351
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01019351