Abstract
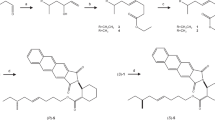
A short synthesis of [4,5-3H2] (E)-9-oxo-2-decenoic acid (ODA), a high-specific-activity tritium-containing isotopomer of the queen bee pheromone, is described. Catalytic tritiation of the ketal of ethyl 9-oxo-4-decenoate introduces tritium into two positions, one of which is completely unactivated. Subsequent transformation by selenation, oxidation, and hydrolysis affords the labeled 9-ODA at >60 Ci/mmol. The material is suitable for biochemical studies of binding and catabolism in ovarian, antennal, and other target tissues.
Similar content being viewed by others
References
Butler, C.G. 1957. The control of ovarian development in worker honeybees (Apis mellifera).Experientia 13:256–257.
Butler, C.G., andGibbons, D.A. 1959. The inhibition of queen rearing by feeding queenless worker honeybees (Apis mellifera) with an extract of “queen substance”.J. Insect Physiol. 2:61–64.
Butler, C.G., Callow, R.K., andJohnson, N.C. 1961. The isolation and synthesis of queen substance, 9-oxo-dec-trans-2-enoic acid, a honeybee pheromone.Proc. R. Soc. Ser. B 155:417–432.
Butler, C.G., Callow, R.K., Greenway, A.R., andSimpson, J. 1974. Movement of the pheromone, 9-oxo-dec-2-enoic acid applied to the body surfaces of honeybees (Apis mellifera).Entomol. Exp. Appl. 17:112–116.
Clement, W.H., andSelwitz, C.M. 1964. Improved procedure converting higher ω-olefins to methyl ketones with palladium chloride.J. Org. Chem. 29:241–243.
Ding, Y.-S., andPrestwich, G.D. 1986. Metabolic transformations of tritium-labeled pheromone by tissues ofHeliothis virescens moths.J. Chem. Ecol. 12:411–429.
Evans, E.A., Warrell, D.C., Elvidge, J.A., andJones, J.R. 1985. Handbook of Tritium NMR Spectroscopy and Applications. John Wiley & Sons, New York.
Gary, N.B. 1962. Chemical mating attractants in the queen honey bee.Science 136:773–774.
Hata, G., Takahashi, K., andMujaka, A. 1971. Palladium-catalyzed reaction of 1,3-dienes with active méthylene compounds.J. Org. Chem. 36:2116–2123.
Johnston, N.C., Law, J.H., andWeaver, N. 1965. Metabolism of 9-keto-dec-2-enoic acid by worker honeybees (Apis mellifera).Biochemistry 4:1615–1621.
Prestwich, G.D. 1987a. Chemistry of hormone and pheromone metabolism in insects.Science 237:999–1006.
Prestwich, G.D. 1987b. Chemical studies of pheromone reception and catabolism,in G. D. Prestwich and G.J. Blomquist (eds.). Pheromone Biochemistry. Academic Press, New York. pp. 473–527.
Prestwich, G. D., Vogt, R.G., andRiddiford, L.M. 1986. Binding and hydrolysis of radiolabeled pheromone and several analogs by male-specific antennal proteins of male mothAntheraea polyphemus.J. Chem. Ecol. 12:323–333.
Prestwich, G.D.,Vogt, R.G., andDing, Y.-S. 1987a. Chemical studies of pheromone catabolism and reception,in J.H. Law (ed.). Molecular Entomology. New York. pp. 57–66.
Prestwich, G.D., andStreinz, L. 1988. Haloacetate analogs of pheromones: Effects on catabolism and electrophysiology inPlutella xylostella.J. Chem. Ecol. 14:1003–1021.
Prestwich, G.D.,Graham, S.M.,Kuo, J.-W., andVogt, R.G. 1987b. Tritium-labeled enantiomers of disparlure: Synthesis and in vitro metabolism. In preparation.
Sharpless, K.B., Lauer, R.F., andTeranishi, A.Y. 1973. Electrophilic and nucleophilic organoselenium reagents. New routes to α, β-unsaturated carbonyl compounds.J. Am. Chem. Soc. 95:6137–6139.
Still, W.C., Kahn, M., andMitra, A. 1978. Rapid Chromatographie technique for preparative separations with moderate resolution.J. Org. Chem. 43:2923–2925.
Tsuji, J.,Masaska, K., andTakahashi, T. 1977. Simple synthesis of queen substance from the butadiene telomer.Tetrahedron Lett. 2267–2268.
Vogt, R.G., Riddiford, L.M., andPrestwich, G.D. 1985. Kinetic properties of a sex pheromone-degrading enzyme: The sensillar esterase ofAntheraea polyphemus.Proc. Natl. Acad. Sci. U.S.A. 82:8827–8831.
Author information
Authors and Affiliations
Additional information
Fellow of the Alfred P. Sloan Foundation (1981–1985) and Camille and Henry Dreyfus Teacher-Scholar (1981–1986).
Rights and permissions
About this article
Cite this article
Webster, F.X., Prestwich, G.D. Synthesis of carrier-free tritium-labeled queen bee pheromone. J Chem Ecol 14, 957–962 (1988). https://doi.org/10.1007/BF01018786
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01018786