Synopsis
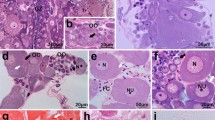
Sections of ovary from plains viscacha, cat, ferret, rabbit, rat, guinea-pig and roe deer have been histochemically processed to demonstrate acetylcholinesterase (AChE) and butyrylcholinesterase (BuChE) in nervous and non-nervous tissue. The effects of different reproductive states on enzyme activity were observed in some animals. AChE-containing nerves were sparse in rabbit and rat but plentiful in cat and roe deer. Nerves containing BuChE were not detectable in ferret or guinea-pig and were rare in cat. Species variations in the activity and type of enzyme were also found in non-neuronal tissues. Some blood vessels in the ovaries of guinea-pig and viscacha contained AChE. No other species showed a reaction for AChE in non-neuronal stromal tissue but BuChE was present at this site in all animals except rat. Granulosa cells reacted for AChE only in cat and rabbit while luteal cells were reactive in cat, rabbit and roe deer. Some BuChE activity was present in granulosa and or luteal cells in all species except roe deer. In rat, BuChE activity in luteal cells increased during oregnaney and the early phase of pseudopregnancy. The difficulty of assigning a function to ovarian cholinesterases is discussed.
Similar content being viewed by others
References
Aitken, R. J., Burton, J., Hawkins, J., Kerr-Wilson, R., Short, R. V. &Steven, D. H. (1973). Histological and ultrastructural changes in the blastocyst and reproductive tract of the roe deer,Capreolus capreolus, during delayed implantation.J. Reprod. Fert. 34, 481–93.
Arvy, L. (1960). Contribution à l'histoenzymologie de l'ovaire.Z. Zellforsch. mikrosk. Anat. 51, 406–20.
Augustinsson, K.-B. (1959). Electrophoresis studies on blood plasma esterases. I. Mammalian plasmata.Acta chem. scand. 13, 571–92.
Bell, C. (1972). Autonomic nervous control of reproduction: circulatory and other factors.Pharmac. Rev. 24, 657–736.
Bell, C. & Malcolm, S. J. (1976). Mechanism of loss of adrenergic fluorescence from vasomotor nerves of the uterus during pregnancy.Proc. Aust. Physiol. Pharmac. Soc. 7, 105P.
Elatchley, F. R. &Donovan, B. T. (1969). Luteolytic effect of prostaglandin in the guinea-pig.Nature, Lond. 221, 1065–6.
Bruce, N. W. &Hillier, K. (1974). The effect of prostaglandin F2α on ovarian blood flow and corpora lutea regression in the rabbit.Nature, Lond. 249, 176–7.
Bulmer, D. (1965). A histochemical study of ovarian cholinesterases.Acta anat. 62, 254–65.
Cottle, M. K. W. &Silver, A. (1970a). Fluorescent granules in the guinea-pig hypothalamus and their possible relation to neurosecretory substance.Z. Zellforsch. mikrosk. Anat. 103, 559–69.
Cottle, M. K. W. &Silver, A. (1970b). Histochemical demonstration of acetylcholinesterase in the hypothalamus of the female guinea-pig.Z. Zellforsch. mikrosk. Anat. 103, 570–88.
Elbadawi, N. A., Smith, J. C. &Schenk, E. A. (1972). Effect of estrogen on uterine cholinesterases.Fedn Proc. Fedn Am. Socs exp. Biol. 31, 246.
Cerebtzoff, M. A. (1959).Cholinesterases. A Histochemical Contribution to the Solution of some Functional Problems, pp. 154–8. London: Pergamon Press.
Hammond, J. Jr. (1974).The Ferret: some observations on Photoperiod and Gonadal Activity, and their role in seasonal Pelt and Body weight changes; the synergistic effect of oestrogen and progesterone on weight gain; and a comparative study of the Corpus Luteum of the Ferret and the Rabbit, p. 27. Cambridge: John Hammond Jr.
Harrison, F. A., Heap, R. B. &Silver, A. (1974). Cholinesterase activity in the autotransplanted ovary of the sheep.J. Physiol., Lond. 242, 10–11P.
Hebb, C. &Linzell, J. L. (1970). Innervation of the mammary gland. A histochemical study in the rabbit.Histochem. J. 2, 491–505.
Hedqvist, P. (1976). Further evidence that prostaglandins inhibit the release of noradrenaline from adrenergic nerve terminals by restriction of availability of calcium.Br. J. Pharmac. 58, 599–603.
Jacobowitz, D. &Wallach, E. E. (1967). Histochemical and chemical studies of the autonomic innervation of the ovary.Endrocinology 81, 1132–9.
Lewis, P. R. (1961). The effect of varying the conditions in the Koelle technique.Biblthca anat. 2, 11–20.
McCracken, J. (1971). Prostaglandin F2α and corpus luteum regression.Ann. N.Y. Acad. Sci. 180, 456–69.
Mendel, B. &Myers, D. K. (1955). Identification of pseudocholinesterase in the tissues of ruminants.Nature, Lond,176, 783–4.
Owman, C. &Sjöberg, N.-O. (1966). Adrenergic nerves in the female genital tract of the rabbit. With remarks on cholinesterase-containing structures.Z. Zellforsch. mikrosk. Anat. 74, 182–97.
Perry, J. S. (1971).The Ovarian Cycle of Mammals. Edinburgh: Oliver & Boyd.
Osengren, E. &Sjöberg, N.-O. (1967). The adrenergic nerve supply to the female reproductive tract of the cat.Am. J. Anat. 121, 271–84.
Silver, A. (1963). A histochemical investigation of cholinesterases at neuromuscular junctions in mammalian and avian muscle.J. Physiol., Lond. 169, 386–93.
Silver, A. (1974).The Biology of Cholinesterases. Amsterdam: North-Holland Publishing Co.
Silver, A. (1977). Acetylcholinesterase in blood vessels of the guinea-pig ovary during different phases of the reproductive cycle.Histochem. J. 9, 341–55.
Skaer, R. J. (1973). Acetylcholinesterase in human erythroid cells.J. Cell Sci. 12, 911–23.
Weir, B. J. (1971). The reproductive physiology of the plains viscacha,Lagostomus maximus.J. Reprod. Fert. 25, 355–63.
Zajicek, J. &Datta, N. (1953). Investigation on the acetylcholinesterase activity of erythrocytes, platelets and plasma in different animal species.Acta haemat. 9, 115–21.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Silver, A. Species variation in the distribution of cholinesterases in the ovary of the plains viscacha, cat, ferret, rabbit, rat, guinea-pig and roe deer. Histochem J 10, 79–102 (1978). https://doi.org/10.1007/BF01003416
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01003416