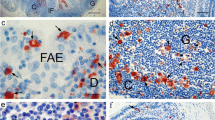
Summary
A new technique of quantitative histochemistry has been developed to study the cellular composition of the follicle-associated epithelium of the mouse Peyer's patch. This technique involves applying naphthol AS-BI phosphate to the surface of intact tissue where it is hydrolysed by alkaline phosphatase present in the luminal membrane of the epithelial cells. Naphthol AS-BI produced by this reaction is then coupled to Fast Red TR diazonium salt at the site of hydrolysis. M cells present in the epithelium contain little alkaline phosphatase activity and, therefore, remain white. Treatment with Alcian Blue is finally used to label goblet cells. Subsequent quantitative analysis of alkaline phosphatase-rich cells is carried out by scanning microdensitometry. Using this technique it is possible to detect two populations of alkaline phosphatase-containing cells in mice reared in a normal animal house environment.
These results are discussed in relation to possible interactions taking place between enteric antigens and the gut-associated lymphoid tissue which could reduce the ability of follicle-associated enterocytes to express alkaline phosphatase.
Similar content being viewed by others
References
Burstone, M. S. (1961) Histochemical demonstration of phosphatases in frozen sections with naphthol AS-phosphates.J. Histochem. Cytochem 9, 146–53.
Bye, W. A., Allan, C. H. &Trier, J. S. (1984) Structure, distribution, and origin of M cells in Peyer's patches of mouse ileum.Gastroenterology 86, 789–801.
Gomori, G. (1939) Microtechnical demonstration of phosphatase in tissue sections.Proc. Soc. Exp. Biol. Med. 42, 23–6.
Gossrau, R. (1980) Conventional techniques for membrane-bound enzymes.Ciba Symp. 73, 67–76.
Gutschmidt, S., Lange, U. &Riecken, E. O. (1980) Kinetic characterization of unspecific alkaline phosphatase at different villus sites of rat jejunum.Histochemistry 69, 189–202.
Kenny, A. J. &Maroux, S. (1982) Topology of microvillar membrane hydrolases of kidney and intestine.Physiol. Rev. 62, 91–128.
Lojda, Z., Gossrau, R. &Schiebler, T. H. (1979)Enzyme Histochemistry: A Laboratory Manual. Berlin: Springer.
Owen, R. L. &Bhalla, D. K. (1983) Cytochemical analysis of alkaline phosphatase and esterase activities and of lectin-binding and anionic sites in rat and mouse Peyer's patch M cells.Am. J. Anat. 168, 199–212.
Owen, R. L. &Jones, A. L. (1974) Epithelial cell specialization within human Peyer's patches: an ultrastructural study of intestinal lymphoid follicles.Gastroenterology 66, 189–203.
Pearse, A. G. E. (1968)Histochemistry Theoretical and Applied, Vol. 1, 3rd edn. London: Churchill.
Smith, M. W. (1985a) Expression of digestive and absorptive function in differentiating enterocytes.Ann. Rev. Physiol. 47, 247–60.
Smith, M. W. (1985b) Selective expression of brush border hydrolases by mouse Peyer's patch and jejunal villus enterocytes.J. Cell. Physiol. 124, 219–25.
Smith, M. W., James, P. S. &Tivey, D. R. (1987) M cell numbers increase after transfer of SPF mice to a normal animal house environment.Am. J. Pathol. 128, 385–9.
Smith, M. W. &Peacock, M. A. (1980) “M” cell distribution in follicle-associated epithelium of mouse Peyer's patch.Am. J. Anat. 159, 167–75.
Ugolev, A. M., Delaey, P., Iezuitova, N. N., Rakhimov, K. R., Timofeeva, N. M. &Stepanova, A. T. (1979) Membrane digestion and nutrient assimilation in early development.Ciba Symp. 70, 221–43.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Smith, M.W., James, P.S., Tivey, D.R. et al. Automated histochemical analysis of cell populations in the intact follicle-associated epithelium of the mouse Peyer's patch. Histochem J 20, 443–448 (1988). https://doi.org/10.1007/BF01002430
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01002430