Abstract
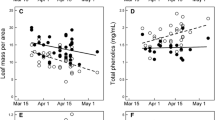
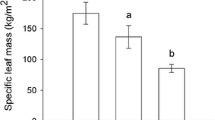
Perennial plants are thought to respond to partial or complete defoliation by producing new foliage that is less susceptible to herbivores because of induction of allelochemicals. Here, I tested this hypothesis by manually removing primary foliage from branches ofQuercus emoryi (Fagaceae) at two different times in the season and monitoring changes in protein and tannin levels and the amount of herbivory relative to control branches. New, secondary leaves had 2.5 × greater hydrolyzable tannin content than mature foliage of control branches. Condensed tannins, which constitute a relatively low fraction of leaf mass, were lower, while protein content was temporarily greater, in new secondary leaves relative to mature leaves. Despite large increases in hydrolyzable tannins, herbivory levels were greater on refoliated branches than on control branches. New foliage is susceptible to herbivory regardless of when it is produced in the season, possibly because lower toughness and higher water content override any induced or developmentally related changes in allelochemistry. My results do not support the hypothesis that postherbivore changes in phytochemistry protect perennial plants from future herbivory, at least within a growing season.
Similar content being viewed by others
References
Auerbach, M.J., andSimberloff, D. 1985. Responses of leaf miners to atypical leaf production patterns.Ecol. Entomol. 9:361–367.
Baldwin, I.T. 1990. Herbivory simulations in ecological research.Trends Ecol. Evol. 5:91–93.
Baldwin, I.T., andSchultz, J.C. 1983. Rapid changes in tree leaf chemistry induced by damage: Evidence for communication between plants.Science 221:277–279.
Barbosa, P., andLetourneau, D.K. (eds.). 1988. Novel Aspects of Insect-Plant Interactions. Wiley-InterScience, New York.
Bate-Smith, E.C. 1977. Astringent tannins ofAcer species.Phytochemistry 16:1421–1426.
Broadhurst, R.B., andJones, W.T. 1978. Analysis of condensed tannins using acidified vanillin.J. Sci. Food Agric. 29:788–794.
Bryant, J.P., Heitkonig, I., Kuropat, andOwen-Smith, N. 1991. Effects of severe defoliation on the long-term resistance to insect attack and on leaf chemistry in six woody species of the southern African savanna.Am. Nat. 137:50–63.
Faeth, S.H. 1985. Quantitative defense theory and patterns of feeding by oak insects.Oecologia (Berlin) 68:34–40.
Faeth, S.H. 1986. Indirect interactions between temporally-separated herbivores mediated by the host plant.Ecology 67:479–494.
Faeth, S.H. 1987. Community structure and folivorous insect outbreaks: the roles of vertical and horizontal interactions, pp. 135–171,in P., Barbosa and J.C., Schultz (eds.). Insect Outbreaks. Academic Press, New York.
Faeth, S.H. 1988. Plant-mediated interactions between seasonal herbivores: enough for evolution or coevolution? pp. 391–414,in K.C., Spencer (ed.). Chemical Mediation of Coevolution. Academic Press, New York.
Faeth, S.H. 1990. Aggregation of a leafminer,Cameraria sp. nov. (Davis): Consequences and causes.J. Ánim. Ecol. 59:569–586.
Faeth, S.H. 1991a. Inducible responses and interactions among oak folivores, pp. 293–324,in M.J. Raupp and D.W. Tallamy (eds.). Phytochemical Induction by Herbivores. Wiley-Interscience, New York.
Faeth, S.H. 1991b. Effects of oak leaf size on abundance, dispersion, and survival of the leafminerCameraria sp. (Lepidoptera: Gracillariidae).Environ. Entomol. 20:196–204.
Faeth, S.H. 1992. Interspecific and intraspecific interactions via plant responses to folivory: An experimental field test.Ecology In press.
Feeny, P. 1970. Seasonal changes in oak leaf tannins and nutrients as a cause of spring feeding by winter moth caterpillars.Ecology 51:565–581.
Fowler, S.V., andLawton, J.H. 1985. Rapidly induced defences and talking trees: the devil's advocate position.Am. Nat. 126:181–195.
Hunter, M.D. 1987. Opposing effects of spring defoliation on late season oak caterpillars.Ecol. Entomol. 12:373–382.
Karban, R. 1991. Inducible resistance in agricultural systems, pp. 402–420,in D.W. Tallamy and M.J. Raupp (eds.). Phytochemical Induction by Herbivores. Wiley-Interscience, New York.
Karban, R., andMyers, J.H. 1989. Induced plant responses to herbivory.Annu. Rev. Ecol. Syst. 20:331–348.
Kearney, T.H., andPeebles, R.H. 1960. Arizona Flora. University of California Press, Berkeley, California.
Neuvonen, S., andHaukioja, E. The effects of inducible resistance in host foliage on birch-feeding herbivores, pp. 277–292,in D.W. Tallamy and M.J. Raupp (eds.). Phytochemical Induction by Herbivores. Wiley-Interscience, New York.
Pase, C.P., andJohnson, R.R. 1968. Flora and vegetation of the Sierra Ancha experimental forest, Arizona.USDA For. Serv. RM-41:1–19.
Pullin, A.S. 1987. Changes in leaf quality following clipping and regrowth ofUrtica dioica, and consequences for a specialist insect herbivore,Agalis urticae Am. Zool. 49:39–45.
Rockwood, L.L. 1974. Seasonal changes in the susceptibility ofCrescentia alata leaves to the flea beetle,Oedionychus sp.Ecology 55:142–148.
SchultZ, J.C. 1988. Plant responses induced by herbivores.Trends Ecol. Evol. 3:45–49.
Schultz, J.C., andBaldwin, I.T. 1982. Oak leaf quality declines in response to defoliation by gypsy moth larvae.Science 217:149–151.
Silva-Bohorquez, I. 1986. Interspecific interactions between insects on oak trees, with special reference to defoliators and the oak aphid. University of Oxford, DPhil. thesis.
Spencer, K.C. (ed.) 1988. Chemical Mediation of Coevolution. Academic Press, San Diego.
Tallamy, D.W., andRaupp, M.J. (eds.). 1991. Phytochemical Induction by Herbivores. Wiley-Interscience, New York.
Zucker, W.V. 1983. Tannins: Does structure determine function?Am. Nat. 121:335–365.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Faeth, S.H. Do defoliation and subsequent phytochemical responses reduce future herbivory on oak trees?. J Chem Ecol 18, 915–925 (1992). https://doi.org/10.1007/BF00988332
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00988332