Abstract
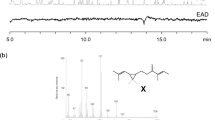
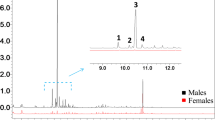
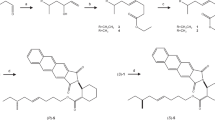
A pheromone mixture containing enantiomerically pure (−)-α-multistriatin of known absolute configuration prepared by total synthesis was found to be as attractive as the natural pheromone to the smaller European elm bark beetle,Scolytus multistriatus, a vector of Dutch elm disease. Its (+)-enantiomer, on the other hand, was no more active than controls in both laboratory and field tests, and at high levels it appeared to inhibit the response to the (−)-enantiomer.
Similar content being viewed by others
References
Borden, J.H., Chong, L., McLean, J.A., Slessor, K.N., andMori, K. 1976.Gnathotrichus sulcatus: Synergistic response to enantiomers of the aggregation pheromone sulcatol.Science 192:894–896.
Cernigliaro, G.J., andKocienski, P.J. 1977. A synthesis of (−)-α-multistriatin.J. Org. Chem. 42:3622–3624.
Elliott, W.J., andFried, J. 1976a. Maytansinoids. Synthesis of a fragment of known absolute configuration involving chiral centers C-6 and C-7.J. Org. Chem. 41:2469–2475.
Elliott, W.J., andFried, J. 1976b. Stereocontrolled synthesis of α-multistriatin, an essential component of the aggregation pheromone for the European elm bark beetle.J. Org. Chem. 41:2475–2476.
Gore, W.E., Pearce, G.T., andSilverstein, R.M. 1975. Relative stereochemistry of multistriatin (2,4-dimethyl-5-ethyl-6,8-dioxabicyclo[3.2.1]octane).J. Org. Chem. 40:1705–1708.
Gore, W.E., andArmitage, I.M. 1976. Lanthanide-induced chemical shifts and the relative stereochemistry of multistriatin (2,4-dimethyl-5-ethyl-6,8-dioxabicyclo[3.2.1]octane).J. Org. Chem. 41:1926–1930.
Lanier, G.N., Gore, W.E., Pearce, G.T., Peacock, J.W., andSilverstein, R.M. 1977. Response of the European elm bark beetle,Scolytus multistriatus (Coleoptera: Scolytidae), to isomers and components of its pheromone.J. Chem. Ecol. 3:1–8.
Lanier, G.N., Silverstein, R.M., andPeacock, J.W. 1976. Attractant pheromone of the European elm bark beetle (Scolytus multistriatus): Isolation, identification, synthesis, and utilization studies, pp. 149–175, J.F. Anderson and H.K. Kaya(eds.). Perspectives in Forest Entomology. Academic Press, New York.
Moeck, H.A. 1970. An olfactometer for the bioassay of attractants for scolytids.Can. Entomol. 102:792–796.
Mori, K. 1976. Pheromone synthesis. Synthesis of (1S,2R,4S,5R)-(−)-α-multistriatin the pheromone in the smaller European elm bark beetleScolytus multistriatus.Tetrahedron 32:1979–1981.
Peacock, J.W., Cuthbert, R.A., Gore, W.E., Lanier, G.N., Pearce, G.T., andSilverstein, R.M. 1975. Collection on Porapak Q of the aggregation pheromone ofScolytus multistriatus (Coleoptera: Scolytidae).J. Chem. Ecol. 1:149–160.
Pearce, G.T., Gore, W.E., Silverstein, R.M., Peacock, J.W., Cuthbert, R.A., Lanier, G.N., andSimeone, J.B. 1975. Chemical attractants for the smaller European elm bark beetleScolytus multistriatus (Coleoptera: Scolytidae).J. Chem. Ecol. 1:115–124.
Pearce, G.T., Gore, W.E., andSilverstein, R.M. 1976. Synthesis and absolute configuration of multistriatin.J. Org. Chem. 41:2797–2803.
Tumlinson, J.H., Klein, M.G., Doolittle, R.E., Ladd, T.L., andProveaux, A.T. 1977. Identification of the female Japanese beetle sex pheromone: Inhibition of male response by an enantiomer.Science 197:789–792.
Vité, J.P., Klimetzek, D., Loskant, G., Hedden, R., andMori, K. 1976. Chirality of insect pheromones: Response interruption by inactive antipodes.Naturwissenschaften 63:582–583.
Wood, D.L., Browne, L.E., Ewing, D., Lindahl, K., Bedard, W.D., Tilden, P.E., Mori, K., Pitman, G.B., andHughes, P.R. 1976. Western pine beetle: Specificity among enantiomers of male and female components of an attractant pheromone.Science 192:896–898.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Elliott, W.J., Hromnak, G., Fried, J. et al. Synthesis of multistriatin enantiomers and their action onScolytus multistriatus (Coleoptera:Scolytidae). J Chem Ecol 5, 279–287 (1979). https://doi.org/10.1007/BF00988242
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00988242