Abstract
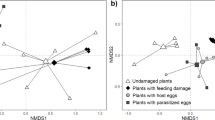
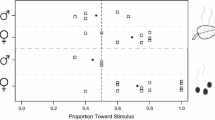
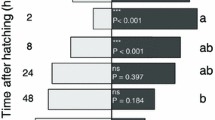
Recently parasitoids were hypothesized to encounter a reliability-detectability problem relating to chemical stimuli from the first and second trophic level, when searching for hosts. The relative role of infochemicals originating from the host,Pieris brassicae (second trophic level), and its food plant, cabbage (first trophic level), have been investigated with respect to long-range host location by the larval parasitoidCotesia glomerata. Flight-chamber dual choice tests showed that uninfested cabbage plants are least attractive to female wasps. Host larvae and their feces were more attractive than clean plants but far less attractive than artificially damaged and herbivore-damaged plants. The plant-host complex, with host larvae actively feeding on the plant, was the most attractive odor source for the parasitoids. The data indicate that one of the solutionsC. glomerata uses to solve the reliability-detectability problem is to respond to infochemicals that are emitted from herbivore-damaged plants. Whether these infochemicals are herbivore-induced synomones that are produced by the plant remains to be demonstrated. Infochemicals emitted by the herbivore or its by-products are of little importance in the foraging behavior ofC. glomerata.
Similar content being viewed by others
References
Dicke, M. 1988. Prey preference of the phytoseiid miteTyphlodromus pyri: 1. Response to volatile kairomones.Exp. Appl. Acarol. 4:1–13.
Dicke, M., andSabelis, M.W. 1988. How plants obtain predatory mites as bodyguards.Neth. J. Zool. 38:148–165.
Dicke, M., andTakabayashi, J. 1991. Specificity of induced indirect defence of plants against herbivores.Redia 74(3)Appendix: 105–113.
Dicke, M., Jong, M. de, Alers, M.P.T., Stelder, F.C.T., Wunderink, R., andPost, J. 1989. Quality control of mass-reared arthropods: Nutritional effects on performance of predatory mites.J. Appl. Entomol. 108:462–475.
Dicke, M.,Baarlen, P.Van, Wessels, R., andDŸkman, H. 1993. Herbivory induces systemic production of plant volatiles that attract the herbivore's predators: extraction of endogenous elicitor.J. Chem. Ecol. 19 (in press).
Dicke, M., Beek, T.A. van, Posthumus, M.A., Ben Dom, N., Bokhoven, H. van, andGroot, Ae. de 1990a. Isolation and identification of volatile kairomone that affects acarine predatorprey interactions. Involvement of host plant in its production.J. Chem. Ecol. 16:381–396.
Dicke, M., Sabelis, M.W., Takabayashi, J., Bruin, J., andPosthumus, M.A. 1990b. Plant strategies of manipulating predator-prey interactions through allelochemicals: prospects for application in pest control.J. Chem. Ecol. 16:3091–3118.
Ding, D., Swedenborg, P.D., andJones, R.L. 1989. Plant odor preferences and learning inMacrocentrus grandii (Hymenoptera: Braconidae), a larval parasitoid of the European corn borer,Ostrinia nubilalis (Lepidoptera: Pyralidae).J. Kans. Entomol. Soc. 62:164–176.
Eller, F.J., Tumlinson, J.H., andLewis, W.J. 1988a. Beneficial arthropod behavior mediated by airborne semiochemicals. VII Source of volatiles mediating the host-location flight behavior ofMicroplitis croceipes (Cresson) (Hymenoptera: Braconidae), a parasitoid ofHeliothis zea (Boddie) (Lepidoptera: Noctuidae).Environ. Entomol. 17:745–753.
Eller, F.J., Tumlinson, J.H., andLewis, W.J. 1988b. Beneficial arthropod behavior mediated by airborne semiochemicals. II. Olfactometric studies of host location by the parasitoidMicroplitis croceipes (Cresson) (Hymenoptera: Braconidae).J. Chem. Ecol. 14:425–434.
Elst, I.F.A.M., Vernede, R., andPak, G.A. 1991. Response of the parasitoid waspCotesia glomerata to odour cues from the host-habitat complex.Proc. Exp. Appl. Entomol. 2:109–114.
Elzen, G.W., Williams, H.J., andVinson, S.B. 1983. Response by the parasitoidCampoletis sonorensis (Hymenoptera: Ichneumonidae) to chemicals (synomones) in plants: implications for host habitat location.Environ. Entomol. 12:1873–1877.
Elzen, G.W., Williams, H.J., andVinson, S.B. 1984. Role of diet in host selection ofHeliothis virescens by parasitoidCampoletis sonorensis (Hymenoptera: Ichneumonidae).J. Chem. Ecol. 10:1535–1541.
Kaiser, L., andCardé, R.T. 1992. In-flight orientation to volatiles from the plant-host complex inCotesia rubecula (Hym.: Braconidae): increased sensitivity through olfactory experience.Physiol. Entomol. 17:62–67.
Keller, M.A. 1990. Responses of the parasitoidCotesia rubecula to its hostPieris rapae in a flight tunnel.Entomol. Exp. Appl. 57:243–249.
Kitano, H. 1978. Studies on the ovipositing activity and host-searching behavior ofApanteles glomeratus L. (Hymenoptera, Braconidae), a parasitoid on the cabbage white butterfly.Kontyu Tokyo 46:152–161.
Laing, J.E., andLevin, D.B. 1982. A review of the biology and bibliography ofApanteles glomeratus (L.) (Hymenoptera: Braconidae).Biocontrol News Information 3:7–23.
Lewis, W.J., Nordlund, D.A., Gueldner, R.C., Teal, P.E.A., andTumlinson, J.H. 1982. Kairomones and their use for management of entomophagous insects. XIII. Kairomonal activity forTrichogramma spp. of abdominal tips, excretion, and a synthetic sex pheromone blend ofHeliothis zea (Boddie) moths.J. Chem. Ecol. 8:1323–1331.
McAuslane, H.J., Vinson, S.B., andWilliams, H.J. 1991. Stimuli influencing host microhabitat location in the parasitoidCampoletis sonorensis.Entomol. Exp. Appl. 58:267–277.
Mizell, R.F., III, Frazier, J.L., andNebeker, T.E. 1984. Response of the clerid predatorThanasimus dubius (F.) to bark beetle pheromones and tree volatiles in a wind tunnel.J. Chem. Ecol. 10:177–187.
Monteith, L.G. 1964. Influence of the health of food plant of the host on host-finding by tachinid parasites.Can. Entomol. 96:1477–1482.
Nealis, V.G. 1986. Responses to host kairomones and foraging behaviour of the insect parasiteCotesia rubecula (Hymenoptera: Braconidae).Can. J. Zool. 64:2393–2398.
Nettles, W.C. 1980. AdultEucelatoria sp.: Response to volatiles from cotton and okra plants and from larvae ofHeliothis virescens, Spodoptera eridania, andEstigmene acrea.Environ. Entomol. 9:759–763.
Noldus, L.P.J.J., Lenteren, J.C. van, andLewis, W.J. 1991. HowTrichogramma parasitoids use moth sex pheromones as kairomone: orientation behaviour in a wind tunnel.Physiol. Entomol. 16:313–327.
Nordlund, D.A., Jones, R.L., andLewis, W.J. (eds.). 1981. Semiochemicals, Their Role in Pest Control. Wiley & Sons, New York, 306 pp.
Roth, J.P., King, E.G., andHensley, S.D. 1982. Plant, host, and parasite interactions in the host selection sequence of the tachinidLixophaga diatraeae.Environ. Entomol. 11:273–277.
Sabelis, M.W., Afman, B.P., andSlim, P.J. 1984. Location of distant spider mite colonies byPhytoseiulus persimilis: localization and extraction of a kairomone.Acarology 6 (1):431–440.
Sato, Y. 1979. Experimental studies on parasitization byApanteles glomeratus. IV. Factors leading a female to the host.Physiol. Entomol. 4:63–70.
Sokal, R.R., andRohlf, F.J. 1981. Biometry. W.H. Freeman & Company, New York.
Steinberg, S., Dicke, M., Vet, L.E.M., andWanningen, R. 1992. Response of the braconid parasitoidCotesia (= Apanteles) glomerata to volatile infochemicals: Effect of bioassay setup, parasitoid age and experience and barometric flux.Entomol. Exp. Appl. 63:163–175.
Takabayashi, J., Dicke, M., andPosthumus, M. A. 1991a. Variation in composition of predatorattracting allelochemicals emitted by herbivore-infested plants: relative influence of plant and herbivore.Chemoecology 2:1–6.
Takabayashi, J., Dicke, M., andPosthumus, M.A. 1991b. Induction of indirect defence against spider-mites in uninfested lima bean leaves.Phytochemistry 30:1459–1462.
Turlings, T.C.J., andTumlinson, J.H. 1992. Systemic release of chemical signals by herbivoreinjured corn.Proc. Natl. Acad. Sci. U.S.A. 89:8399–8402.
Turlings, T.C.J., Scheepmaker, J.W.A., Vet, L.E.M., Tumlinson, J.H., andLewis, W.J. 1990a. How contact foraging experiences affect preferences for host-related odors in the larval parasitoidCotesia marginiventris (Cresson) (Hymenoptera: Braconidae).J. Chem. Ecol. 16:1577–1589.
Turlings, T.C.J., Tumlinson, J.H., andLewis, W.J. 1990b. Exploitation of herbivore-induced plant odors by host-seeking parasitic wasps.Science 250:1251–1253.
Turlings, T.C.J., Tumlinson, J.H., Eller, F.J., andLewis, W.J. 1991a. Larval-damaged plants: Source of volatile synomones that guide the parasitoidCotesia marginiventris to the microhabitat of its hosts.Entomol. Exp. Appl. 58:75–82.
Turlings, T.C.J., Tumlinson, J.H., Heath, R.R., Proveaus, A.T., andDoolittle, R.E. 1991b. Isolation and identification of allelochemicals that attract the larval parasitoid,Cotesia marginiventris (Cresson), to the microhabitat of one of its hosts.J. Chem. Ecol. 17:2235–2251.
Ullyet, G.C. 1953. Biomathematics and insect population problems.Entomol. Soc. South Afr. Mem. 2:1–89.
Vet, L.E.M. 1985. Olfactory microhabitat location in some eucoilid and alysiine species (Hymenoptera), larval parasitolds of Diptera.Neth. J. Zool. 35:720–730.
Vet, L.E.M., andDicke, M. 1992. Ecology of infochemical use by natural enemies of herbivores in a tritrophic context.Annu. Rev. Entomol. 37:141–172.
Vité, J., andWilliamson, D.L. 1970.Thanasimus dubius: prey perception.J. Insect Physiol. 16:233–239.
Vinson, S.B. 1976. Host selection by insect parasitoids.Annu. Rev. Entomol. 21:109–134.
Vinson, S.B. 1981. Habitat location, pp. 51–77,in D.A. Nordlund, R.L. Jones, and W.J. Lewis (eds.). Semiochemicals: Their Role in Pest Control. Wiley, New York.
WÄckers, F.L., andLewis, W.J. 1992. Olfactory and visual learning and their interaction in host site location byMicroplitis croceipes. Biol. Control In press.
Weseloh, R.M. 1981. Host location by parasitoids, pp. 79–95,in D.A. Nordlund, R.L. Jones, and W.J. Lewis (eds.). Semiochemicals: Their Role in Pest Control. Wiley, New York.
Wiskerke, J.S.C., andVet, L.E.M. 1991. Comparison of twoCotesia species foraging for solitarily and gregariously feedingPieris host species.Proc. Exp. Appl. Entomol. 2:190–195.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Steinberg, S., Dicke, M. & Vet, L.E.M. Relative importance of infochemicals from first and second trophic level in long-range host location by the larval parasitoidCotesia glomerata . J Chem Ecol 19, 47–59 (1993). https://doi.org/10.1007/BF00987470
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00987470