Conclusions
-
1.
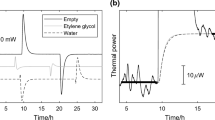
The suggested design for a calorimetric apparatus serves, to evaluate quickly with a small error (±3.5%) the specific heat of liquids.
-
2.
This measurement precision can be attained even with a temperature difference between the calorimetric liquid and the jacket attaining 80°C, provided that the equipment comprises a vessel with vacuum-filled walls, a cork dust stopper, an ebonite carrier with a glass tube, etc.
-
3.
It has been established experimentally that the thermal value of the suggested calorimetric equipment remains virtually constant for a calorimetric liquid temperature change from 20 to 95°C.
Similar content being viewed by others
Literature cited
M. M. Popov, Thermometry and Calorimetry [in Russian], Izd. MGU, Moscow (1954).
Additional information
Translated from Izmeritel'naya Tekhnika, No. 5, pp. 51–53, May, 1968.
Rights and permissions
About this article
Cite this article
Kovalenko, B.M. Calorimeter for determining the specific heat of liquids. Meas Tech 11, 639–642 (1968). https://doi.org/10.1007/BF00986629
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00986629