Abstract
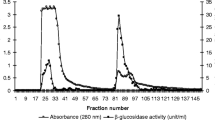
β-Glucosidase activity has been determined in homogenate and in centrifugation fractions of 7-day-old and adult rat brain; maximum activity was found at pH 4 and pH 5. Of the adult brain, more than 50% of the activity was concentrated in the 800-g sediment fraction (P1), while in the brain of 7-day-old rat about 20% was found in the corresponding fraction. The activity maximum in all fractions after a 2% Triton X-100 treatment occurs at pH 5. Addition of Triton to adult brain homogenate enhances the activity, but this stimulation is less than the sum of the activities observed at pH 4 and pH 5 in the absence of Triton. Triton addition to brain homogenate of 7-day-old rat results in a fall in activity at pH 4 and in a maximum at pH 5. In rat brain homogenate subjected to sonication, a loss of activity is observed at pH 4, scarcely at pH 5; the activity loss is completely abolished and turned into an increase under the influence of Triton. This increase equals the level obtained when Triton is added to an untreated brain homogenate. Sonication of rat brain homogenate leads to changes in the distribution pattern; about 25% of the activity of the adult brain is found in the P1 fraction compared to 50% in the corresponding fraction of the untreated brain. Fractionation of a sonicated brain homogenate from adult rat reveals that at pH 4 most activity (52%) is concentrated in the 20,000-g pellet (P2), 23% in supernatant fluid (S2); at pH 5 the opposite is observed: most activity (49%) is found in the 20,000-g supernatant (S2) and 23% in the 20,000-g pellet (P2). In the presence of Triton the activity of the sonicated brain homogenate of adult rat increases; this stimulation roughly equals the sum of the corresponding activities measured at pH 4 and pH 5 in the absence of Triton.
Similar content being viewed by others
References
Asp, N. G. 1970. Rat intestinal β-galactosidase. Biochem. J. 117:369.
Asp, N. G. 1971. Human small intestinal β-galactosidase. Biochem. J. 121:299.
Baccino, F. M., Rita, G. A., andZuretti, M. F. 1971. Studies on the structure-bound sedimentability of some rat liver lysosome hydrolases. Biochem. J. 122:363.
Beck, C., andTappel, A. L. 1968. Rat liver lysosomal β-glucosidase: A membrane enzyme. Biochim. Biophys. Acta 151:159.
Beutler, E., andKuhl, W. 1970. The diagnosis of the adult type of Gaucher's disease and its carrier state by demonstration of deficiency of β-glucosidase activity in peripheral blood leukocytes. J. Lab. Clin. Med. 76:747.
Brady, R. O., Gal, A. E., Kanfer, J. N., andBradley, R. M. 1965. The metabolism of glucocerebrosides. III. Purification and properties of a glucosyl- and galacyosylceramide-cleaving enzyme from rat intestinal tissue. J. Biol. Chem. 240:3766.
Carubelli, R., andFulsiani, D. R. P. 1971. Neuraminidase activity in brain and liver of rats during development. Biochim. Biophys. Acta 237:78.
Conchie, J., andHay, A. J. 1963. Mammalian glycosidases. 4. The intracellular localization of mannosidase,N-acetyl-glucosaminidase and fucosidase in mammalian tissues. Biochem. J. 87:354.
De Duve, C., Pressman, B. C., Gianetto, R., Wattiaux, R., andAppelmans, F. 1955. Tissue fractionation studies. 6. Intracellular distribution patterns of enzymes in rat liver tissue. Biochem. J. 60:604.
Gatt, S., andRapport, M. M. 1966. Isolation of β-galactosidase and β-glucosidase from brain. Biochim. Biophys. Acta 113:567.
Hermans, I. J., Hoving, H., andHooghwinkel, G. J. M. 1973. β-Glucosidases in developing rat brains. Neurobiology 3:45.
Ho, M. W., andO'Brien, J. S. 1971. Gaucher's disease: Deficiency of ‘acid’ β-glucosidase and reconstruction ofenzyme activity in vitro. Proc. Natl. Acad. Aci. U.S.A. 68:2810.
Hultberg, B., andÖckerman, P. A. 1970. β-Glucosidase activities in human tissue. Findings in Gaucher's disease. Clin. Chim. Acta 28:169.
Lisman, J. J. W., Veltkamp, W. A., andHooghwinkel, G. J. M. 1971. Action of Triton X-100 on the extraction and activation of glycosidases from bovine brain tissue. Neurobiology 1:121.
Lisman, J. J. W., andHooghwinkel, G. J. M. 1973. Human brain β-glucosidase and β-galactosidase. Influence of Triton X-100 on the extraction, activation and gel chromatographic behaviour. Neurobiology 3:264.
Mellors, A., andTappel, A. L. 1967. Hydrolysis of phospholipids by a lysosomal enzyme. J. Lipid Res. 8:479.
Patel, A., andKoenig, H. 1976. Brain lysosomal hydrolases: 1. Solubilization and electrophoretic behavior of acid hydrolases in nerve-ending and mitochondrial-lysosomal fractions from rat brain. Effects of autolysis, neuraminidase and storage. Neurochem. Res. 1:275.
Pugh, D., Leaback, D. H., andWalker, P. G. 1957.N-Acetyl-β-glucosaminidase in rat tissues preparations. Biochem. J. 65:16p.
Rahman, Y. E., Verhagen, J., andWiel, D. F. M. 1970. Evidence of a membrane-bound phospholipase A in rat liver lysosomes. Biochem. Biophys. Res. Commun. 38:670.
Robins, E., Hirsch, E., andEmmons, S. S. 1968. I Assay, some properties, and distribution of β-galactosidase, β-glucuronidase, and β-glucosidase. J. Biol. Chem. 243:4246.
Robinson, D., Jordan, J. W., andHorsburgh, T. 1972. TheN-acetyl-β-d-hexosaminidases of calf and human brain. J. Neurochem. 19:1975.
Ugazio, G., andPani, P. 1963. Differential release of bound hydrolases from rat liver lysosomes treated with a non-ionic surface active substance. Exp. Cell Res. 31:424.
Vanier, M. T., Holm, M., Ohman, R., andSvennerholm, L. 1971. Developmental profiles of gangliosides in human and rat brains. J. Neurochem. 18:581.
Weismann, B., Rowin, G., Marshall, J., andFriederici, D. 1967. Mammalian α-acetyl glucosaminidase. Enzymic properties, tissue distribution and intracellular localization. Biochemistry 6:207.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hermans, I.J. β-d-glucosidase in fractions from rat brain. Neurochem Res 3, 711–724 (1978). https://doi.org/10.1007/BF00965994
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00965994