Abstract
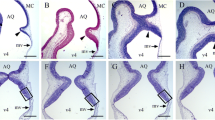
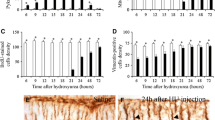
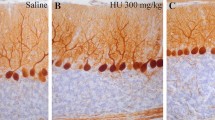
The effect of methylnitrosourea (MNU) on cerebellar and cerebral DNA, RNA, protein, lysosomal enzymes (acid DNase, RNase, phosphatase, and beta-glucuronidase), and 2′,3′-cyclic nucleotide 3′-phosphohydrolase (2′,3′-CNPase) activities was studied in rats from birth through 12 days of age. Subcutaneous injection of MNU in a dose of 0.625 mmol/kg caused a suppression of increase in weights and content of DNA, RNA, and protein of cerebellum, but no changes in those of the cerebrum or in body weight. Ratios of protein and RNA to DNA were substantially elevated by MNU in the cerebellum but not in the cerebrum.
Acid DNase and acid RNase activities of MNU-treated rats were significantly elevated beyond the increase of these activities in controls in the cerebellum, but no change in these activities by MNU was observed in the cerebrum. A slight elevation in acid phosphatase activity was observed in the cerebellum but not in the cerebrum after MNU pretreatment. Beta-glucuronidase and 2′,3′-CNPase activities were not changed in the cerebellum or in the cerebrum. These results suggest that in the developing brain, especially in the cerebellum at the mitotic stage, MNU caused cell damage and inhibited cell mitosis.
Similar content being viewed by others
References
Alexandrov, W. A. 1969. Transplacental blastomogenic action of N-nitrosomethylurea in rat offspring. Problem of oncology 15:55–61.
Allfrey, V. G., andMirsky, A. E. 1952. Some aspects of the deoxyribonuclease activities of animal tissues. J. Gen. Physiol. 36:227–241.
Balazs, R., Kovacs, S., Teichgraber, R., Cocks, W. A., andEayrs, J. T. 1968. Biochemical effects of thyroid deficiency on the developing brain. J. Neurochem. 15:1335–1349.
Bosch, D. A., Gerrits, P. O., andEbels, E. J. 1972. The cytotoxic effect of ethylnitrosourea and methylnitrosourea on the nervous system of the rat at different stages of development. Z. Krebsforsch 77:308–318.
Bosch, D. A., Ipema, A., andEbels, E. J. 1973. Different cytotoxic effects of methyl- and ethylnitrosourea on developing rat tissues and the influence of cycloheximide on cell death. Z. Krebsforsch 79:255–266.
Bosch, D. A. 1977. Short and long term effects of methyl-and ethylnitrosourea (MNU & ENU) on the developing nervous system of the rat. Acta Neurol. Scand. 55:106–122.
Burton, K. 1956. A study of the colorimetric estimation of deoxyribonucleic acid. Biochem. J. 62:315–323.
Druckrey, H., Ivankovic, S., undPreussman, R. 1965. Selektive Erzeugung maligner Tumoren im Gehirn und Rückenmark von Ratten durch N-Methyl-N-nitrosoharnstoff. Z. Krebsforsch. 66:389–408.
Druckrey, H., Ivankovic, S., andGimmy, J. 1973. Cancerogene Wirkung von Methyl- und Äthylnitrosoharnstoff (MNH und ÄNH) nach einmaliger intracerebraler bzw. intracarotidaler injektion bei neugeborenen und jungen BD-Ratten. Z. Krebsforsch. 79:282–297.
Druckrey, H., Preussman, R., andIvakovic, S. 1969. N-nitroso compounds in organotropic and transplacental carcinogenesis. Ann. N. Y. Acad. Sci. 163:676–696.
Garrett, E. R., Goto, S., andStubbins, J. F. 1965. Kinetics of solvolyses of various N-alkyl-N-nitrosoureas in neutral and alkaline solutions. J. Pharm. Sci. 54:119–123.
Ittel, M. E., andMandel, P. 1977. Nuclear ribonuclease activities of rat brain during postnatal development. J. Neurochem. 28:1355–1358.
Kurihara, T., andTsukada, Y. 1967. The regional and subcellular distribution of 2′-3′-cyclic nucleotide 3′-phosphohydrolase in the central nervous system. J. Neurochem. 14:1167–1174.
Kurihara, T., Nussbaum, J. L., andMandel, P. 1970. 2′,3′-cyclic nucleotide 3′-phosphohydrolase in brains of mutant mice with deficient myelination. J. Neurochem. 17:993–997.
Lehman, I. R. 1967. Deoxyribonucleases: their relation to deoxyribonucleic acid synthesis. Ann. Rev. Biochem. 36:645–668.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., andRandell, R. J. 1951. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193:265–275.
Matthieu, J. M., Quarles, R. H., De F. Webster, H., Hogan, E. L., andBrady, R. O. 1974. Characterization of the fraction obtained from the CNS (central nervous system) of Jimpy mice by a procedure for myelin isolation. J. Neurochem. 23:517–523.
McDonald, M. R. 1955. Ribonucleases methods in enzymology. Meth. Enzymol. 2:427–447.
McCalla, D. R., Renver, A., andKitar, R. 1968. Inactivation of biologically active N-methyl-N-nitroso compounds in aqueous solution: effect of various conditions of PH and illumination. Canad. J. Biochem. 46:807–811.
Mejbaum, W. 1939. Estimation of small amounts of pentose especially in derivatives of adenylic acid. Z. Physiol. Chem. 258:117–120.
Mirault, M. E., andScherrer, K. 1972. In vitro processing of Hela cell peribosomes by a nucleolar endoribonuclease. Fed. Eur. Biochem. Soc. Lett. 20:233–238.
Rabié, A., Matrat, M. S., Clavel, M. C., Clos, J., andLegrand, J. 1977. Effects of methylazoxymethanol given at different stages of postnatal life on development of the rat brain: Comparison with those of thyroid deficiency. J. Neurobiol. 8:337–354.
Robins, E., Hirsch, H. E., andEmmons, S. S. 1968. Glycosidases in the nervous system. J. B. C. 243:4246–4252.
Schreiber, D., Jänisch, W., Scholtze, P., andTausch, H. 1968. Experimentelle Tumoren des Zentralnervensystems. Naturwissenschaft 55:495.
Schmidt, G., andThannhauser, S. J. 1945. A method for the determination of deoxyribonucleic acid, ribonucleic acid and phosphoproteins in animal tissues. J. Biol. Chem. 161:83–89.
Shirivastaw, K. P. andSubba Rao, K. 1975. Changes in the levels of DNA, RNA, protein and DNases in developing and old chick brain. J. Neurochem. 25:861–865.
Sprinkle, T. J., Zaruba, M. E., andMckhann, G. M. 1978. Activity of 2′,3′-cyclic nucleotide 3′-phosphodiesterase in regions of rat brain during development. J. Neurochem. 30:309–314.
Subba Rao, K. 1973. Acid deoxyribonuclease activity in developing rat brain. Life Sci. 12:89–96.
Sung, S. C. 1971. Thymidine kinase in the developing rat brain. Brain Res., 35:268–271.
Swann, P. F. 1968. The rate of breakdown of methyl methanesulphonate, dimethyl sulphonate and N-methyl-N-nitrosourea in the rat. Biochem. J. 110:49–52.
Wasterlein, C. G., andPlum, F. 1973. Vulnerability of developing rat brain to electroconvulsive seizures. Arch. Neurol. 29:38–45.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fujimori, K., Sunouchi, M., Inoue, K. et al. Cytotoxic effects of methylnitrosourea on developing brain. Neurochem Res 8, 193–206 (1983). https://doi.org/10.1007/BF00963920
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00963920