Abstract
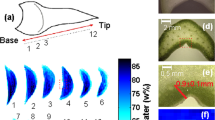
Differences in water binding were measured in the leaf cells ofMesembryanthemum crystallinum L. plants grown under high-salinity conditions by using nuclear-magnetic-resonance (NMR) imaging. The 7-Tesla proton NMR imaging system yielded a spatial resolution of 20·20·100 μm3. Images recorded with different spin-echo times (4.4 ms to 18 ms) showed that the water concentrations in the bladder cells (located on the upper and lower leaf surface), in the mesophyll cells and in the water-conducting vessels were nearly identical. All of the water in the bladder cells and in the water-conducting vessels was found to be mobile, whilst part of the water in the mesophyll cells was bound. Patches of mesophyll cells could be identified which bound water more strongly than the surrounding mesophyll cells. Optical investigations of leaf cross-sections revealed two types of mesophyll cells of different sizes and chloroplast contents. It is therefore likely that in the small-sized mesophyll cells water is strongly bound. A long-term asymmetric water exchange between the mesophyll cells and the bladder cells during Crassulacean acid metabolism has been described in the literature. The high density of these mesophyll cells in the lower epidermis is a possible cause of this asymmetry.
Similar content being viewed by others
Abbreviations
- CAM:
-
Crassulacean acid metabolism
- NMR:
-
nuclear magnetic resonance
- TE :
-
spin-echo time
References
Aguayo, J., Blackband, S., Schoeniger, J., Mattingly, M., Hintermann, M. (1986) Nuclear magnetic resonance imaging of a single cell. Nature322, 190–191
Bloch, F. (1946) Nuclear induction. Phys. Rev.70, 460–474
Bottomley, P.A., Foster, T.H., Argersinger, R.E., Pfeifer, L.H. (1984) A review of normal tissue hydrogen NMR relaxation times and relaxation mechanisms from 1–100 MHz: Dependence on tissue type, NMR frequency, temperature, species, excision, and age. Med. Phys.11, 425–448
Bottomley, P.A., Rogers, H.H., Foster, T.H. (1986) NMR imaging shows water distribution and transport in plant root systems in situ. Proc. Natl. Acad. Sci. USA83, 87–89
Connelly, A., Lohman, J.A.B., Loughman, B.C., Quiquampoix, H., Ratcliffe, R.G. (1987) High resolution imaging of plant tissues by NMR. J. Exp. Bot.38, 1713–1723
Eccles, C.D., Callaghan, P.T. (1986) High-resolution imaging. The NMR microscope. J. Magn. Reson.68, 393–398
Engmann, K.F. (1934) Studien über die Leistungsfähigkeit der sukkulenter Pflanzen. Beih. Bot. Cbl.52a, 381–414
Fullerton, G.D., Potter, J.L., Dornbluth, N.C. (1982) NMR relaxation of protons in tissues and other macromolecular water solutions. Magn. Reson. Imag.1, 209–228
Haberlandt, G. (1918) Physiologische Pflanzenanatomie. W. Engelmann Verlag, Leipzig
Hüsken, D., Steudle E., Zimmermann, U. (1978) Pressure probe technique for measuring water relations of cells in higher plants. Plant Physiol.61, 158–163
Johnson, G.A., Brown, J., Kramer, P.J. (1987) Magnetic resonance microscopy of changes in water content in stems of transpiring plants. Proc. Natl. Acad. Sci. USA84, 2752–2755
Kramer, D. (1979) Ultrastructural observations on developing leaf bladder cells ofMesembryanthemum crystallinum L.. Flora168, 193–204
Lauterbur, P.C. (1973) Image formation by induced local interactions: Examples employing nuclear magnetic resonance. Nature242, 190–191
Lüttge, U., Fischer, E., Steudle, E. (1978) Membrane potential and salt distribution in epidermal bladders and photosynthetic tissue ofMesembryanthemum crystallinum. Plant Cell Environ.1, 121–129
Mansfield, P., Morris, P.G. (1982) NMR imaging in biomedicine. Academic Press, New York
Metcalfe, C.R., Chalk, L. (1950) Ficoidaceae. Anatomy of the dicotyledons, vol. I, pp. 706–711. Claredon Press, Oxford
Oberstein, O. (1913) Über den Bau der Blattspitzen der Mesembrianthema-Barbata. Beih. Bot. Cbl.29, 298–302
Rygol, J., Büchner, K.-H., Winter, K., Zimmermann, U. (1986) Day night variations in turgor pressure in individual cells ofMesembryanthemum crystallinum L.. Oecologia69, 171–175
Rygol, J., Zimmermann, U., Balling, A. (1989) Water relations of individual leaf cells ofMesembryanthemum crystallinum at high and low external salinity. J Membr. Biol.107, 203–212
Schuhmacher, J.H., Clorius, J.H., Semmler, W., Hauser, H., Matys, E.R., Maier-Borst, W., Hull, W.E. (1987) NMR relaxation times T1 and T2 of water in plasma from patients with long carcinoma: Correlation of T2 with blood sedimentation rate. Magn. Reson. Med.5, 537–547
Stejskal, E.O., Tanner, J.E. (1965) Spin diffusion measurements: Spin echoes in the presence of a time-dependent field gradient. J. Chem. Phys.42, 288–292
Steudle, E., Jeschke, W.D. (1983) Water transport in barley roots. Planta158, 237–248
Steudle, E., Lüttge, U., Zimmermann, U. (1975) Water relations of the epidermal bladder cells of the halophytic speciesMesembryanthemum crystallinum: Direct measurements of hydrostatic pressure and hydraulic conductivity. Planta126, 229–246
Steudle, E., Zimmermann, U. (1971) Hydraulische Leitfähigkeit vonValonia utricularis. Z. Naturforsch.26b, 1302–1311
Winter, K. (1973) CO2-Fixierungsreaktionen bei der SalzpflanzeMesembryanthemum crystallinum unter variierten Außenbedingungen. Planta114, 75–85
Winter, K. (1985) Crassulacean acid metabolism. In: Photosynthetic mechanisms and the environment, pp. 329–387, Barber, J., Baker, N.R., eds. Elsevier, Amsterdam
Zimmermann, U. (1978) Physics of turgor- and osmoregulation. Annu. Rev. Plant Physiol.29, 121–148
Zimmermann, U. (1988) Determination of water relations of plant cells by means of the pressure probe technique. Methods Enzymol. (in press)
Zimmermann, U., Hüsken, D., Schulze, E.D. (1980) Direct turgor pressure measurements in different leaf cells ofTradescantia virginiana. Planta149, 445–453
Zimmermann, U., Raede, H., Steudle, E. (1969) Kontinuierliche Druckmessung in Pflanzenzellen. Naturwissenschaften56, 634–635
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Walter, L., Balling, A., Zimmermann, U. et al. Nuclear-magnetic-resonance imaging of leaves ofMesembryanthemum crystallinum L. plants grown at high salinity. Planta 178, 524–530 (1989). https://doi.org/10.1007/BF00963822
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00963822