Conclusions
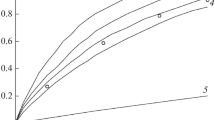
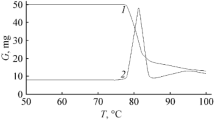
The kinetics of the thermal decomposition of N-methyl-, N-t-butyl-, and N-neopentyl-N′-methoxydiazene-N-oxides have been studied in the gas phase. The reactions are first order with activation parameters: E 204, 148, and 206 kJ/mole, log(A/sec−1) 14.5, 13.3, and 14.6, respectively. A mechanism has been proposed for the decomposition of the MeN- and t-BuCH2N-derivatives by the primary homolytic rupture of the N-OMe bond, and in the case of the t-BuN-analog, via a five-membered cyclic transition state with migration of a β-H to the N atom.
Similar content being viewed by others
Literature cited
M. V. George, R. W. Kierstead, and G. F. Wright, Can. J. Chem.,37, 679 (1957).
I. N. Zyuzin and D. B. Lempert, Izv. Akad. Nauk SSSR, Ser. Khim., 831 (1985).
D. L. Marquard, J. STAM,11, 431 (1963).
V. V. Dubikhin, G. M. Nazin, and G. B. Manelis, Izv. Akad. Nauk SSSR, Ser. Khim., 1339 (1971).
V. V. Dubikhin, G. M. Nazin, D. N. Sokolov, and G. B. Manelis, Izv. Akad. Nauk SSSR, Ser. Khim., 1416 (1971).
L. Batt, R. T. Mibne, and R. D. McCulloch, Int. J. Chem. Kinet.,9, 567 (1977).
W. Forst, J. Chem. Phys.,44, 2349 (1966).
L. O. Atovmyan, N. I. Golovina, and I. N. Zyuzin, Izv. Akad. Nauk SSSR, Ser. Khim., 1309 (1987).
G. M. Nazin, Usp. Khim.,41, 1537 (1972).
V. N. Grebennikov, G. B. Manelis, G. M. Nazin, et al, Izv. Akad. Nauk SSSR, Ser. Khim., 1721 (1984).
P. S. Engel, Chem. Revs.,80, 99 (1980).
J. W. Lown, S. M. S. Chauchan, R. R. Koganty, and A.-M. Sapse, J. Am. Chem. Soc.,106, 6401 (1984).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 7, pp. 1506–1509, July, 1988.
Rights and permissions
About this article
Cite this article
Zyuzin, I.N., Lempert, D.B. & Nechiporenko, G.N. Kinetics of thermal decomposition of n-alkyl-n′-methoxydiazene-n-oxides in the gas phase. Russ Chem Bull 37, 1329–1332 (1988). https://doi.org/10.1007/BF00962732
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00962732