Conclusions
-
1.
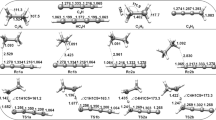
A calculation of the potential energy surfaces (PES's) for the reductive cleavage of the C-X bond in the radical anions of the methyl halides CH3X∸ has been carried out by the semiempirical MNDO method with consideration of the effect of a solvent in the point-dipole model.
-
2.
The solvent appreciably modifies the form of the PES's of the radical anions in comparison to the form calculated for the gaseous phase. The calculations attest to the formation of stationary states for X=F and Br, but they do not indicate the formation of stable particles for X=Cl. The profile of the PES for X=I is characterized by a deep minimum with a barrier in the direction of dissociation equal to ∼1 eV.
Similar content being viewed by others
Literature cited
E. Canadell, P. Karafiloglou, and L. Salem, J. Am. Chem. Soc.,102, 855 (1980).
T. Clark, J. Chem. Soc. Chem. Commun., 515 (1981).
C. P. Andrieux, I. Gallardo, J. M. Saveant, and K. B. Su, J. Am. Chem. Soc.,108, 638 (1986).
K. Ya. Burshtein, J. Mol. Struct. (Theochem),153, 195 (1987).
J. Chandrasekhar, S. F. Smith, and W. L. Jorgensen, J. Am. Chem. Soc.,107, 154, 2474 (1985).
J. D. Madura and W. L. Jorgensen, J. Am. Chem. Soc.,108, 2517 (1986).
M. J. S. Dewar and W. Thiel, J. Am. Chem. Soc.,99, 4899 (1977).
W. J. Hehre and J. A. Pople, J. Am. Chem. Soc.,92, 2191 (1970).
Handbook of Molecular Constants of Inorganic Compounds [in Russian], Khimiya, Leningrad (1979), p. 279.
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 9, pp. 2051–2055, September, 1988.
Rights and permissions
About this article
Cite this article
Burshtein, K.Y., German, É.D. Quantum-chemical study of the cleavage of the C-X bonds in the radical ions CH3X∸ (X=F, Cl, Br, I). Russ Chem Bull 37, 1837–1841 (1988). https://doi.org/10.1007/BF00962499
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00962499