Conclusions
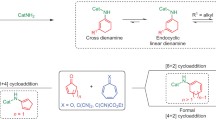
The homodimerization of spiro{bicyclo[2.2.1]hepta-2,5-diene-7,1′-cyclopropane} (I) has been investigated in the presence of Ni-, Fe-, Co-, Rh-, and Ir-based metal complex catalysts. Depending on the nature of the metal and its ligand environment cyclodimerization of (I) can take place via [2π+2π]-, [2π+2π+2π]-, and [4π+4π]-cycloaddition schemes, to give high yields of hepta-, octa-, and nonacyclic strained hydrocarbons.
Similar content being viewed by others
Literature cited
U. M. Dzhemilev, R. I. Khusnutdinov, Z. S. Muslimov, et al., Izv. Akad. Nauk SSSR, Ser. Khim., 2299 (1981).
S. R. Rafikov, U. M. Dzhemilev, R. I. Khusnutdinov, et al., Izv. Akad. Nauk SSSR, Ser. Khim., 902 (1982).
U. M. Dzhemilev, R. I. Khusnutdinov, and G. A. Tolstikov, Usp. Khim.,56, 65 (1987).
Patent No. 1197083 (FRG); RZhKhim 6N75 (1967).
U.S. Patent No. 4207080; Chem. Abstr.,93, 204160 (1980).
R. I. Khusnutdinov, V. A. Dokichev, A. A. Panasenko, et al., Izv. Akad. Nauk SSSR, Ser. Khim., 2492 (1984).
K. Alder, H.-J. Ache, and F. H. Flock, Chem. Ber.,93, 1888 (1960).
E. W. Abel, M. A. Bennet, and G. Wilkinson, J. Chem. Soc., 3181 (1959).
R. Colton, R. H. Fating, and J. E. Knapp, Austr. J. Chem.,23, 1351 (1970).
J. Bowell and B. L. Shaw, J. Chem. Soc., A,3, 583 (1968).
G. Winkhaus and M. Singer, Chem. Ber.,99, 3610 (1966).
K. Wrieze, J. P. Colleman, C. T. Sears, and M. Kubota, Inorg. Synth.,11, 101 (1968).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 11, pp. 2592–2599, November, 1988.
Rights and permissions
About this article
Cite this article
Dzhemilev, U.M., Khusnutdinov, R.I., Muslimov, Z.S. et al. Homodimerization of spiro{bicyclo[2.2.1]Hepta-2,5-Diene-7,1′-cyclopropane}, catalyzed by group VIII transition metal complexes. Russ Chem Bull 37, 2337–2343 (1988). https://doi.org/10.1007/BF00959891
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00959891