Summary
-
1.
Fused KF·2H2O is proposed for the stationary phase of GC analysis of aqueous solutions of polar compounds with an inert gas to be used as the mobile phase, saturated with H2O at ∼20°C.
-
2.
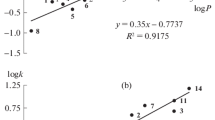
It has been shown that fused KF·2H2O used as a stationary phase effects the separation of polar compounds chiefly by van der Waals interactions, since the normal order of elution of the n-alkanes is observed and the change in the values of the retention indices in the homological series of n-alcohols is described by the general equation.
-
3.
The ability of KF·2H2O for different types of intermolecular interaction were described on the basis of the energy equivalents of the retention indices of five test-substances.
Similar content being viewed by others
Literature cited
V. R. Alishoev, V. G. Berezkin, S. N. Viktorova, V. S. Gavrichev, and V. S. Fateeva, “Chromatographic methods and their use in the petrochemical and petroleum refining industries,” in: Abstracts of the 1st International Conference of the Countries of CMEA, Bratislava (1982), p. 31.
R. V. Golovnya, I. L. Zhuravleva, and B. M. Polanuer, Izv. Akad. Nauk SSSR, Ser. Khim., (1983), p. 2060.
R. V. Golovnya and A. L. Samusenko, Izv. Akad. Nauk SSSR, Ser. Khim., 2060 (1981).
W. O. McReynolds, Gas-Chromatographic Retention Data, Preston Technical Abs., Evanston (1966), p. 1.
R. V. Golovnya and D. N. Grigoryevna, Chromatographia, 17, 613 (1983).
R. V. Golovnya and T. A. Misharina, Usp. Khim.,171 (1980).
W. R. Supina, The Packed Column in Gas Chromatography, Supelco, Beliefonte, PA (1977).
R. V. Golovnya, Chromatographia,12, 533 (1979).
R. V. Golovnya and D. N. Grigoreva (Grigoryeva), Chromatographia,18, 449 (1984).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 2, pp. 348–352, February, 1987.
Rights and permissions
About this article
Cite this article
Polanuer, B.M., Golovnya, R.V., Zhuravleva, I.L. et al. Crystal hydrate of potassium fluoride as a new stationary phase for gas chromatographic analysis of aqueous solutions of polar substances. Russ Chem Bull 36, 307–310 (1987). https://doi.org/10.1007/BF00959370
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00959370