Conclusions
-
1.
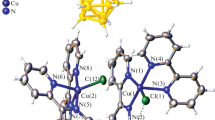
It was found that the final product of dissolution of metallic copper in a tetrahydrofuran solution of o-quinone in an excess of LiCl is a copper(II) biscatecholate complex.
-
2.
A scale of the electrode potentials characterizing the individual redox stages in the process of dissolution of copper in the conditions studied was elaborated; the scale permits predicting the capacity of copper and its cations and Q,\(SQ^{\underset{\raise0.3em\hbox{$\smash{\scriptscriptstyle-}$}}{ \bullet } }\) and Cat2− for mutual redox transformations in tetrahydrofuran.
Similar content being viewed by others
Literature cited
G. A. Abakumov, V. A. Muraev, and G. A. Razuvaev, Dokl. Akad. Nauk SSSR,215, 1113 (1974).
G. A. Abakumov, V. K. Cherkasov, A. V. Lobanov, and G. A. Razuvaev, Izv. Akad. Nauk SSSR, Ser. Khim., 1610 (1984).
Yu. N. Kukushkin and S. Ch. Dkhara, Zh. Neorg. Khim.,15, 1585 (1970).
G. A. Abakumov, A. V. Lobanov, V. K. Cherkasov, and G. A. Razuvaev, Inorg. Chim. Acta,49, 135 (1981).
G. Bodlender and O. Storbeck, Z. Anorg. Allgem. Chem.,31, 1 (1921).
O. Brado and R. T. Iwamoto, Inorg. Chim. Acta,3, 663 (1969).
S. Arland and J. Rawsthorne, Acta Chem. Scand.,24, 1578 (1970).
K. Sugasaka and A. Fujii, Bull. Chem. Soc. Jpn.,49, 8286 (1976).
F. Gordon and R. Ford, The Chemist's Companion [Russian translation], Mir, Moscow (1976), p. 443.
Yu. V. Karyakin and I. I. Angelov, Pure Chemicals [in Russian], Khimiya, Moscow (1974), p. 240.
H. H. Schulze and W. Flaig, Liebigs Ann. Chem.,575, 231 (1972).
E. Muller, A. Rieker, K. Scheftler, and A. Moosmayer, Angew. Chem.,78, 98 (1966).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 2, pp. 292–296, February, 1987.
Rights and permissions
About this article
Cite this article
Abakumov, G.A., Cherkasov, V.K. & Chechet, Y.V. Study of the dissolution of metallic copper in the presence of 3,5-di-tert-butyl-1,2-benzoquinone and lithium chloride in tetrahydrofuran. Russ Chem Bull 36, 256–260 (1987). https://doi.org/10.1007/BF00959358
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00959358