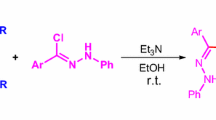
Abstract
For 1-amino- and 1-oxy-Δ2-1,2,3-triazolines, retardation (on the NMR time scale) is observed for rotation around the exocyclic N-N or N-O bond; the measured barriers to topomerization amount to 8.3–13.8 kcal/mole.
Similar content being viewed by others
Literature cited
R. G. Kostyanovskii, G. K. Kadorkina, G. V. Shustov, and K. S. Zakharov, Dokl. Akad. Nauk SSSR,221, 370 (1975).
R. G. Kostyanovskii, A. V. Prosyanik, A. I. Mishchenko, et al., Izv. Akad. Nauk SSSR, Ser. Khim., 1780 (1979).
R. G. Kostyanovskii, A. V. Prosyanik, A. I. Mishchenko, et al., Izv. Akad. Nauk SSSR, Ser. Khim., 882 (1980).
R. G. Kostyanovskii, G. K. Kadorkina, I. I. Chervin, et al., Khim. Geterotsikl. Soedin., 1495 (1980).
A. I. Mishchenko, A. V. Prosyanik, P. N. Belov, et al., Khim. Geterotsikl. Soedin., 338 (1984).
A. V. Prosyanik, P. N. Belov, N. I. Kol'tsov, and V. I. Markov, Zh. Org. Khim.,21, 911 (1985).
G. V. Shustov, A. B. Zolotoi, P. N. Belov, et al., Izv. Akad. Nauk SSSR, Ser. Khim., No. 8, 1820 (1990).
V. G. Dashevskii, Conformations of Organic Molecules [in Russian], Khimiya, Moscow (1974).
G. J. Bishop, B. J. Price, and I. O. Sutherland, J. Chem. Soc, Chem. Commun., 672 (1967).
S. M. Shevchenko, Sovrem. Probl. Org. Khim., No. 6, 162 (1978).
M. Raban and D. Kost, Tetrahedron,40, 3345 (1984).
M. Raban and D. Kost, J. Org. Chem.,37, 499 (1972).
B. J. Price and I. O. Sutherland, J. Chem. Soc, Chem. Commun., 1070 (1967).
G. V. Shustov, N. B. Tavakalyan, N. L. Zaichenko, and R. G. Kostyanovskii, Izv. Akad. Nauk SSSR, Ser. Khim., 2183 (1980).
G. V. Shustov, A. B. Zolotoi, N. L. Zaichenko, et al., Tetrahedron,40, 2151 (1984).
A. Gieren, K. Burger, and J. Fehn, Angew. Chem.,84, 212 (1972).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 4, pp. 790–794, April, 1991.
Rights and permissions
About this article
Cite this article
Zaichenko, N.L., Shustov, G.V., Prosyanik, A.V. et al. Topomerization of substituted 1-amino- and 1-oxy-Δ2-1,2,3-triazolines. Russ Chem Bull 40, 692–696 (1991). https://doi.org/10.1007/BF00958556
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00958556